Depression is one of the most common mental disorders. According to WHO, in 2012, more than 350 million people worldwide suffered from depression. Depression is a leading cause of disability and a significant contributor to the global burden of disease [1]. Despite the variety of antidepressants currently used, it is not always possible to achieve the desired treatment effect. Acute depressive symptoms are relieved in 60-70% of patients, remission can be achieved only in 20-40% [2]. This encourages researchers to look for more effective approaches to treating depressive disorders. In this regard, a drug such as venlafaxine deserves attention.
Venlafaxine appeared in 1994 as one of the first third-generation antidepressants acting on the serotonin and noradrenergic systems of the brain - a “dual” action antidepressant. However, the mechanism of action of venlafaxine actually consists of a “triple” action, since it inhibits the neurotransmitters not only serotonin, norepinephrine, but also dopamine. At a dose of 75-125 mg/day, the drug acts as a serotonin reuptake inhibitor, at a dose of 125-225 mg/day - serotonin and norepinephrine, at a dose of 225-375 mg/day - serotonin, norepinephrine and dopamine [3].
Abroad, venlafaxine and other “dual” action antidepressants are drugs of second choice. They are used in case of minimal or no effect from SSRI treatment for 2-4 weeks [4]. A pronounced anti-anxiety effect of venlafaxine was noted [5–8]. Venlafaxine has been found to be effective and well-tolerated, as well as being more effective than SSRIs in the treatment of severe depression [9, 11]. In terms of achieving remission, venlafaxine is superior to SSRIs and placebo [12-14]. However, there is evidence [15] of equal effectiveness of venlafaxine and escitalopram in the treatment of severe depressive disorders.
It was found that in the treatment of severe depression, the tactic of rapidly increasing the dose of venlafaxine (up to 375 mg/day for 5 days) leads to an earlier onset of clinical effect than when treating with imipramine (up to 200 mg/day for 5 days), and a higher level of therapeutic response by the 3rd week of treatment compared with fluoxetine [16]. Earlier clinical response with high doses of venlafaxine was found in several placebo-controlled studies [17, 18].
In Russia, venlafaxine began to be used in 2005. A number of studies [19–24] have established its effectiveness in mild and moderate depression, psychosomatic disorders, as well as somatic and neurological pathologies. In the treatment of severe depression, there is less effectiveness and a slower onset of the therapeutic effect than in the treatment of moderate depression, and there is no connection between the severity of the therapeutic effect and the daily dose of the drug [25].
The purpose of this study is to identify factors influencing the effectiveness of venlafaxine in the treatment of moderate and severe depression, and on this basis to develop optimal therapeutic approaches.
Material and methods
An observational study examined the use of venlafaxine in real-world clinical practice. 62 patients with severe depression aged from 19 to 61 years were examined. All of them were treated at the Psychiatry Clinic named after. S.S. Korsakov First Moscow State Medical University named after. THEM. Sechenov in 2009-2012.
The study included patients with a depressive episode of moderate to severe severity and recurrent affective disorder, a current episode of depression of moderate severity or severe (categories F32.1, F32.2, F33.1, F33.2 according to ICD-10). At inclusion, the total score on the Montgomery-Åsberg Depression Rating Scale (MADRS) was greater than 25.
Venlafaxine was prescribed as the only antidepressant drug; If necessary, other drugs were added to therapy. The selection of the optimal dose of venlafaxine was carried out individually in accordance with the severity of the mental state and the leading psychopathological syndrome, individual tolerance, and taking into account the somatic condition. Venlafaxine was not prescribed to patients with a history of episodes of mania or hypomania, a high suicidal risk, organic diseases of the central nervous system, acute and chronic psychoses, addiction diseases, severe somatic pathology (uncontrolled arterial hypertension, coronary heart disease, a history of myocardial infarction, increased intraocular pressure, angle-closure form of glaucoma, renal and liver failure).
The main clinical and anamnestic data of the examined patients are presented in Table. 1.
Table 1. Characteristics of the examined patients Note. 1 - average value; in parentheses are the minimum and maximum values; 2 - median; interquartile range in square brackets; 3 - in remission.
The mental state of patients was assessed by clinical, psychopathological and psychometric methods. The psychometric scales used were MADRS, Beck Depression, Clinical Global Impression (CGI-I and CGI-S subscales), and Side Effects Usage (UKU).
Patients were observed during inpatient treatment and subsequently on an outpatient basis until the 12th week of therapy inclusive (84 days).
The average initial dose of venlafaxine was 75 [75; 150] mg1/day, average therapeutic - 225 [150; 375] mg1/day.
The patients' condition was assessed on the day of venlafaxine prescription, on days 3, 7, 10, 14, 21, 28, 42 and 84 of therapy.
The effectiveness of therapy was assessed clinically - by the number of responders (patients whose total score on the MADRS scale decreased by 50% or more). The condition was regarded as remission if the total value on the MADRS scale became less than 12 points. The rate of development of the therapy effect was assessed by the decrease in the total MADRS score, the number of responders and clinical improvement in the condition of patients on a certain day of therapy. Side effects of therapy were identified through a targeted survey of patients and using the UKU scale.
The analysis of the results was carried out using standardized methods of statistical processing of medical data, taking into account the peculiarities of data processing in psychiatry [26].
Venlafaxine Organica, 30 pcs., 37.5 mg, film-coated tablets
Venlafaxine, which itself does not have increased binding to plasma proteins, practically does not increase the concentration of simultaneously taken drugs, which are characterized by high binding to plasma proteins. No clinically significant interaction with antihypertensive (including beta-blockers, ACE inhibitors, diuretics) and antidiabetic drugs was detected.
Caution should be exercised when co-administered with other drugs that affect the central nervous system, since the interaction of venlafaxine with such drugs has not been studied.
MAO inhibitors. The simultaneous use of venlafaxine with MAO inhibitors, as well as within 14 days after their discontinuation, is contraindicated (the risk of severe side effects, including death, is likely). Therapy with MAO inhibitors can be prescribed no less than 7 days after discontinuation of the drug Venlafaxine. Venflaxin should be discontinued at least 7 days before starting reversible selective MAO inhibitors (moclobemide). The weakly reversible and non-selective MAO inhibitor linezolid (an antimicrobial drug) and methylene blue (IV dosage form) are also not recommended for simultaneous use with venlafaxine.
Serotonergic agents. Caution should be exercised in the simultaneous use of drugs that affect the serotonergic transmission of drugs, such as triptans (including sumatriptan, zolmitriptan), SSRIs, SNRIs (prolonged seizures have been reported), tricyclic antidepressants, lithium, sibutramine or fentanyl (including including its analogues - dextromethorphan, tramadol), as well as excess sources of tryptophan due to the increased potential risk of serotonin syndrome.
Alcohol. During treatment with venlafaxine, alcohol should be completely avoided. Alcohol increases the psychomotor dysfunction that venlafaxine can cause.
Lithium. Lithium preparations do not have a significant effect on the pharmacokinetics of venlafaxine.
Diazepam. There was no effect of orally administered diazepam on the pharmacokinetics of venlafaxine and EDV, and, conversely, venlafaxine did not change the pharmacokinetics of diazepam and its metabolite desmethyldiazepam. In addition, the administration of both of these drugs does not impair the psychomotor effects and psychometric indicators caused by diazepam.
Cimetidine. The simultaneous administration of cimetidine and venlafaxine led to a delay in metabolism during the first passage of venlafaxine through the liver. Oral clearance of venlafaxine decreased by 43%, and AUC and Cmax of the drug increased by 60%. However, such an impact was not evident for EFA. Since the total activity of venlafaxine and EFA is expected to increase only slightly, no dose adjustment will be required for most normal patients. However, in patients with existing (detected) hypertension, elderly patients and those with impaired liver or kidney function, the dose of venlafaxine may be adjusted.
Haloperidol. In a study where venlafaxine was administered in the Css stage at a dose of 150 mg/day, a 42% decrease in total oral haloperidol clearance was observed after a 2 mg oral dose; while AUC increased by 70%, and Cmax by 88%, while T1/2 of haloperidol did not change. This should be taken into account when choosing the correct dose of haloperidol.
Imipramine. Venlafaxine does not impair the pharmacokinetics of imipramine and 2-hydroxyimipramine. However, the AUC, Cmax and Cmin of desipramine (the active metabolite of imipramine) increased by approximately 35% when co-administered with venlafaxine. The concentration of 2-hydroxydesipramine also increases 2.5 or 4.5 times (depending on the dose of venlafaxine: 37.5 mg or 75 mg 2 times a day), but the clinical significance of this fact is unknown.
Metoprolol. When using metoprolol and venlafaxine simultaneously, caution should be exercised because due to pharmacokinetic interaction, the concentration of metoprolol in the blood plasma increases by approximately 30-40%, without changing the concentration of its active metabolite alpha-hydroxymetoprolol. The clinical significance of this interaction has not been studied. Metoprolol does not affect the AUC of venlafaxine and EDV.
Risperidone. When used concomitantly with risperidone (despite an increase in the AUC of risperidone), the pharmacokinetics of a pair of active molecules (risperidone and 9-hydroxyrisperidone) do not change significantly when combined with venlafaxine.
Clozapine. During post-marketing studies of venlafaxine, it was found that when used simultaneously with clozapine, its concentration in the blood plasma increases. This was manifested by an increase in the side effects of clozapine, especially in relation to the incidence of seizures.
Indinavir. With simultaneous use, the pharmacokinetics of indinavir changes (AUC decreases by 28% and Cmax decreases by 36%). There are no changes in the pharmacokinetics of venlafaxine. The clinical significance of this fact is unknown.
Ketoconazole. A pharmacokinetic study when combined with ketoconazole showed an increase in plasma concentrations of venlafaxine and EDV in subjects whose initial metabolism by CYP2D6 is either good (X-Met) or poor (P-Met). In particular, the Cmax of venlafaxine increased by 26% for X-Met and by 48% for P-Met. EFA Cmax values increased by 14 and 29% in X-Met and P-Met subjects, respectively. The AUC of venlafaxine increased by 21% in X-Met and by 70% in P-Met. EFA AUC values increased by 23 and 33% in X-Met and P-Met subjects, respectively.
Drugs that affect blood clotting and platelet function (NSAIDs, acetylsalicylic acid preparations and other anticoagulants). Serotonin, released by platelets, plays an important role in hemostasis (stopping bleeding). Epidemiological studies demonstrate an association between the use of psychotropic medications that interfere with serotonin reuptake and the incidence of upper GI bleeding. This relationship is enhanced if NSAIDs, drugs containing acetylsalicylic acid, or other anticoagulants are used simultaneously. The risk of bleeding has been shown to increase when SSRIs and SNRIs (including venlafaxine) are prescribed concomitantly with warfarin. Patients prescribed warfarin should be closely monitored for PT and/or partial thromboplastin time, especially when co-administration with venlafaxine is started or stopped.
Interaction with other drugs at the level of studied metabolism with cytochrome P450 isoenzymes. The main routes of metabolism of venlafaxine include the isoenzymes CYP2D6 and CYP3A4: the first of them converts venlafaxine into its active metabolite EDV, and the second is less important in the metabolism of venlafaxine compared to CYP2D6 and forms the product N-desmethylvenlafaxine with little pharmacological activity. Preclinical studies have shown, and subsequently confirmed clinically, that venlafaxine is a relatively weak inhibitor of CYP2D6. Therefore, even when prescribing drugs that moderately suppress the activity of this enzyme or in the case of treating patients with a genetically determined decrease in CYP2D6 function, no dose adjustment of venlafaxine is required, because the total concentration of the active substance and active metabolite (venlafaxine and EDV) does not change significantly. This characterizes venlafaxine positively when compared with other antidepressants. Caution should be exercised when co-administered with CYP2D6 inhibitors such as quinidine, paroxetine, fluoxetine, haloperidol, perphenazine, levomepromazine, because in this case, venlafaxine may potentially increase plasma concentrations of these CYP2D6 substrates. In combination with drugs that inhibit both enzymes (CYP2D6 and CYP3A4), special caution is required. Such drug interactions have not yet been sufficiently studied and in this case such a combination of drugs is not recommended. In addition, venlafaxine does not suppress the activity of the enzymes CYP3A4, CYP1A2 and CYP2C9, therefore, no significant interaction is observed with drugs such as alprazolam, caffeine, carbamazepine, diazepam, tolbutamide, terfenadine.
Interaction with ketoconazole is described above. CYP3A3/4 inhibitors such as itraconazole and ritonavir can have a similar effect.
Other interactions with various concomitant therapeutic factors and food. When using venlafaxine, special caution should be exercised during electroconvulsive therapy, because There is no experience with the use of venlafaxine in these conditions. A significant effect of different types of food on the absorption of venlafaxine and its subsequent conversion to EDV has not been identified. Foods (usually high in protein, such as hard cheeses, fish roe, turkey), as well as dietary supplements and fitness diets that contain tryptophan, potentially increase the body's production of serotonin, which may increase the serotonergic side effects of venlafaxine.
Preparations containing St. John's wort. Undesirable pharmacodynamic interactions may occur when Venlafaxine is taken simultaneously with the medicinal plant St. John's wort (herb or various preparations made from it); such a combination is not recommended.
There are reports of false-positive results of the immunochromatographic rapid urine test (test strip) for phencyclidine and amphetamines in patients taking venlafaxine, even several days after discontinuation of venlafaxine. This may be explained by the lack of specificity of this test. Only a confirmatory test in a specialized anti-doping laboratory can distinguish venlafaxine from phencyclidine and amphetamines.
According to the data available to date, venlafaxine has not shown itself to be a drug that causes drug abuse or addiction (both in preclinical receptor affinity studies and in clinical practice).
The use of the antidepressant venlafaxine in patients with chronic pain syndrome
About the article
7971
0
Regular issues of "RMZh" No. 20 dated 09/03/2009 p. 1382
Category: Neurology
Authors: Kamchatnov P.R. 1, Radysh B.B. , Kutenev A.V. , Kazakov A.Yu. 1 1 Federal State Autonomous Educational Institution of Russian National Research Medical University named after. N.I. Pirogov Ministry of Health of Russia, Moscow, Russia
For quotation:
Kamchatnov P.R., Radysh B.B., Kutenev A.V., Kazakov A.Yu. Use of the antidepressant venlafaxine in patients with chronic pain syndrome. RMJ. 2009;20:1382.
Chronic pain, which lasts beyond the period required for normal healing and lasts for more than 12 weeks, is a fairly common condition in the population. The results of a population-based study conducted in European countries, which included 50 thousand people, allow us to state that every fifth adult suffers from severe or moderate chronic pain [8]. It turned out that the most common causes of chronic pain syndrome are diseases of the musculoskeletal system - damage to bones, joints, and periarticular tissues. The vast majority of respondents who confirmed the presence of chronic pain receive systematic pain treatment, but more than half of them do not believe that the therapy is completely effective.
Chronic pain, which lasts beyond the period required for normal healing and lasts for more than 12 weeks, is a fairly common condition in the population. The results of a population-based study conducted in European countries, which included 50 thousand people, allow us to state that every fifth adult suffers from severe or moderate chronic pain [8]. It turned out that the most common causes of chronic pain syndrome are diseases of the musculoskeletal system - damage to bones, joints, and periarticular tissues. The vast majority of respondents who confirmed the presence of chronic pain receive systematic pain treatment, but more than half of them do not believe that the therapy is completely effective. A serious problem is neuropathic pain, the occurrence of which is caused by direct damage to the somatosensory system and is not associated with irritation of pain receptors. The results of epidemiological studies suggest that neuropathic pain is experienced by at least 3% of members of the population, although there is evidence of its wider prevalence [25]. According to modern views on the pathophysiology of neuropathic pain, the lesion that causes it can be localized at different levels of the peripheral or central nervous system [1,5]. The developing complex restructuring of the nervous system leads to the formation of a pathological algic system, which ensures the existence of a persistent pain syndrome. An important role in the formation of neuropathic pain syndrome is played by disruption of the functioning of the mechanisms of perception and processing of pain impulses [4]. Of great importance is also the reduction in the activity of the body’s own anti-pain systems, in particular, the pathways emanating from the nuclei of the brain stem (in particular the periaqueductal gray matter), the neurotransmitters in which are serotonin and norepinephrine [19]. Disruption of the functioning of this system is also of great importance for the occurrence of chronic pain syndromes due to various reasons. Considering the complex pathophysiological mechanisms of the formation of chronic pain and neuropathic pain syndromes, painkillers (for example, paracetamol) and non-steroidal anti-inflammatory drugs are not always effective in relieving them. Moreover, their extremely low effectiveness for true neuropathic pain has been established. At the same time, the results of repeated surveys of medical workers indicate that it is non-steroidal anti-inflammatory drugs that are most often prescribed to this group of patients. It should be borne in mind that long-term, often uncontrolled use is associated with a high risk of complications, including severe ones, the most common of which are lesions of the gastric mucosa with the appearance of ulcerations, increased blood pressure, and an increased risk of atherothrombotic complications [8,20 ]. The results of a number of experimental studies and clinical studies indicate that today the optimal approach to treating such patients is the use of antidepressants and antiepileptic drugs. There is sufficient experience in the clinical use of antidepressants in the relief of pain syndromes in patients with neuropathic pain and chronic pain syndromes. For this purpose, tricyclic antidepressants are the most widely used, and the maximum experience gained in randomized clinical trials has been accumulated with amitriptyline. Based on the results of a meta-analysis of 19 randomized clinical trials performed in a double-blind manner (a total of 2515 patients with neuropathic pain syndromes, excluding headaches and migraines were included), it was found that antidepressants are most effective against neuropathic pain syndromes caused by distal diabetic pain polyneuropathy and postherpetic neuralgia. This group of drugs turned out to be less effective against pain syndromes caused by HIV infection and in some other clinical conditions [24]. The authors of the cited meta-analysis, like most other researchers, note that to achieve a therapeutic effect, the use of drugs in high doses is often required, which is associated with an increased risk of side effects. In particular, this complicates the widespread use of drugs in outpatient settings and reduces patient adherence to treatment. For the treatment of patients with neuropathic pain and chronic pain syndromes, in addition to tricyclic antidepressants, drugs from the group of serotonin reuptake inhibitors are also used, which, in addition, when prescribed in therapeutic doses, have the ability to suppress the reuptake of norepinephrine - “dual-action” drugs, a representative of which is venlafaxine (Velafax) [19]. The results of experimental studies convincingly prove that venlafaxine has its own analgesic activity, which is not associated with its antidepressant properties. Thus, the positive effect of venlafaxine, associated with the peculiarities of its chemical structure, is observed in patients with and without chronic pain syndromes associated with depressive disorders [7]. Interestingly, in some cases, the analgesic effect occurs when using doses of the drug that are lower than those that cause the antidepressant effect itself. It is also believed that the analgesic effect in this situation is due to the interaction of the drug with both serotonin and norepinephrine receptors; modulation of the synaptic uptake of neurotransmitters by dopamine may play a significant role. In addition, there is information that the analgesic effect of the drug to a certain extent can be explained by its interaction with opioid systems (primarily with
Venlafaxine (Velaxin) tablets 75 mg No. 30
A country
Russia
The country of production may vary depending on the batch of goods. Please check with the operator for detailed information when confirming your order.
Active substance
Venlafaxine
Compound
1 tablet contains: venlafaxine (in the form of hydrochloride) 75 mg.
Excipients: microcrystalline cellulose 134.62 mg, pregelatinized starch 99.00 mg, colloidal silicon dioxide (aerosil) 1.64 mg, talc 6.60 mg, magnesium stearate 3.30 mg. Flat-cylindrical tablets with a chamfer and a score, white or white with a yellowish tint. Light marbling is allowed.
pharmachologic effect
Venlafaxine is an antidepressant that does not chemically belong to any class of antidepressants (tricyclic, tetracyclic or others) and is a racemate of two active enantiomers. Venlafaxine and its main metabolite, O-desmethylvenlafaxine (ODV), are potent serotonin and norepinephrine reuptake inhibitors (abbreviated as SNRIs or SNRIs) and weak dopamine reuptake inhibitors. The mechanism of antidepressant action is associated with the ability of the drug to enhance the activity of neurotransmitters during the transmission of nerve impulses in the central nervous system (CNS). Venlafaxine and EDV are equally effective in influencing the reuptake of the above-mentioned neurotransmitters, while they do not have an affinity (studied in vitro) for cholinergic (muscarinic), histamine (H1), alpha1-adrenergic, opioid and benzodiazepine receptors, and do not suppress the activity of monoamine oxidase (MAO) . In terms of inhibition of serotonin reuptake, venlafaxine is inferior to selective serotonin reuptake inhibitors (SSRIs).
Indications for use
Depression: prevention and treatment.
Mode of application
Inside. Venlafaxine is taken with food, preferably at the same time, without chewing and with liquid. The recommended starting dose is 75 mg in two divided doses daily (37.5 mg 2 times a day). Depending on tolerability and effectiveness, the dose may be gradually increased to 150 mg/day. If necessary, the dose is increased to 225 mg/day. Dose increases of 75 mg/day can be made at intervals of 2 weeks or more; if clinically necessary, due to the severity of symptoms, it is possible to increase the dose in a shorter period of time, but not less than 4 days. Higher doses (up to a maximum daily dose of 375 mg/day in 2-3 doses) require inpatient monitoring of patients. After achieving the required therapeutic effect, the daily dose can be gradually reduced to the minimum effective level. Maintenance therapy and relapse prevention: Maintenance treatment can last 6 months or more. The minimum effective doses used in the treatment of a depressive episode are prescribed. Renal failure: in case of mild renal failure (glomerular filtration rate (GFR) more than 30 ml/min), no dosage adjustment is required. In case of moderate renal failure (GFR 10-30 ml/min), the dose should be reduced by 25-50%. Due to the prolongation of T1/2 of venlafaxine and its active metabolite (EAM), such patients should take the entire dose once daily. It is not recommended to use venlafaxine in severe renal failure (GFR less than 10 ml/min), since reliable data on such therapy are lacking. During hemodialysis, the daily dose should be reduced by 50%; the drug should be taken after the end of the hemodialysis session. Liver failure: in case of mild liver failure (prothrombin time (PT) less than 14 seconds), no dosage adjustment is required. In case of moderate liver failure (PT 14 to 18 seconds), the daily dose should be reduced by 50% or more. It is not recommended to use venlafaxine in severe liver failure, since reliable data on such therapy are lacking. Elderly patients: the elderly age of the patient in the absence of any acute or chronic diseases does not require a change in dose, however (as when prescribing other drugs) caution is required when treating elderly patients. Elderly patients should use the lowest effective dose. When increasing the dose, the patient should be under close medical supervision. Discontinuation of the drug Discontinuation of the drug should be done gradually to minimize the risk associated with drug withdrawal. With a course of treatment of 6 weeks or more, the period of gradual withdrawal of the drug should be at least 2 weeks and depends on the dose, duration of therapy and the individual characteristics of the patient.
Interaction
Venlafaxine, which itself does not have increased binding to plasma proteins, practically does not increase the concentration of concomitantly taken drugs, which are characterized by high binding to plasma proteins. No clinically significant interaction with antihypertensive (many pharmacological groups, including beta blockers, ACE inhibitors and diuretics) and antidiabetic drugs was detected. Caution should be exercised when co-administered with other drugs that affect the central nervous system, since combinations of venlafaxine with all such drugs have not been studied. MAO inhibitors The simultaneous use of venlafaxine with MAO inhibitors, as well as within 14 days after their discontinuation, is contraindicated (the risk of severe side effects, including death, is likely). Therapy with MAO inhibitors can be prescribed no less than 7 days after discontinuation of the drug Venlafaxine. Venlafaxine should be discontinued at least 7 days before starting reversible selective MAO inhibitors (moclobemide). The weakly reversible and non-selective MAO inhibitor linezolid (an antimicrobial drug) and methylene blue (IV dosage form) are also not recommended for concomitant use with venlafaxine. Serotonergic drugs Caution should be exercised in the simultaneous use of drugs that affect the serotonin system of mediators, such as triptans (sumatriptan, zolmitriptan, etc.), selective serotonin reuptake inhibitors (SSRIs) and SNRIs (prolonged convulsions have been reported), tricyclic antidepressants, lithium, sibutramine or fentanyl (and its analogues dextromethorphan, tramadol, etc.), as well as excess sources of tryptophan due to the increased potential risk of serotonin syndrome. Alcohol During treatment with venlafaxine, alcohol (ethanol) should be completely avoided. Alcohol increases the psychomotor dysfunction that venlafaxine can cause. Lithium Lithium preparations do not have a significant effect on the pharmacokinetics of venlafaxine. Diazepam There was no effect of orally administered diazepam on the pharmacokinetics of venlafaxine and EDV, and, conversely, venlafaxine did not change the pharmacokinetics of diazepam and its metabolite desmethyldiazepam. In addition, the administration of both of these drugs does not impair the psychomotor and psychometric effects caused by diazepam. Cimetidine The simultaneous administration of cimetidine and venlafaxine resulted in a delay in metabolism during the “first pass” of venlafaxine. The oral clearance of venlafaxine decreased by 43%, and the area under the pharmacokinetic curve (AUC) and Cmax for this drug increased by 60%. However, no similar impact was found for EFA. Since the total activity of venlafaxine and EFA is expected to increase only slightly, no dose adjustment will be required for most normal patients. However, in patients with existing (detected) hypertension, elderly patients and those with impaired liver or kidney function, the dose of venlafaxine may be adjusted. Haloperidol In a study where venlafaxine was administered at steady state at a dose of 150 mg/day, a 42% decrease in total oral haloperidol clearance was observed following a 2 mg oral dose; while AUC increased by 70%, and Cmax increased by 88%, while T1/2 of haloperidol did not change. This should be taken into account when choosing the correct dose of haloperidol. Imipramine Venlafaxine does not impair the pharmacokinetics of imipramine and 2-hydroxyimipramine. However, the AUC, Cmax and Cmin of desipramine (the active metabolite of imipramine) increased by approximately 35% when co-administered with venlafaxine. The concentration of 2-hydroxydesipramine also increases from 2.5 to 4.5 times (depending on the dose of venlafaxine: 37.5 mg for 12 hours or 75 mg for 12 hours), but the clinical significance of this fact is not known. Metoprolol When using metoprolol and venlafaxine simultaneously, caution should be exercised, since due to pharmacokinetic interaction, the concentration of metoprolol in the blood plasma increases by approximately 30-40%, without changing the concentration of its active metabolite β-hydroxymetoprolol. The clinical significance of this interaction has not been studied. Metoprolol does not affect the AUC of venlafaxine and EDV. Risperidone When used concomitantly with risperidone (despite an increase in the AUC of risperidone), the pharmacokinetics of a pair of active molecules (risperidone and 9-hydroxyrisperidone) do not change significantly when combined with venlafaxine. Clozapine During post-marketing studies of venlafaxine, it was found that when used simultaneously with clozapine, its concentration in the blood plasma increases. This was manifested by an increase in the side effects of clozapine, especially in relation to the incidence of seizures. Indinavir With simultaneous use, the pharmacokinetics of indinavir changes (AUC decreases by 28% and Cmax decreases by 36%). For venlafaxine, no changes in pharmacokinetics are observed. The clinical significance of this fact is unknown. Ketoconazole A pharmacokinetic study when combined with ketoconazole showed an increase in plasma concentrations of venlafaxine and EDV in subjects with both good (X-Met) and poor (P-Met) metabolism by CYP2D6. In particular, the Cmax of venlafaxine increased by 26% for X-Met and by 48% for P-Met. EFA Cmax values increased by 14% and 29% in X-Met and P-Met subjects, respectively. The AUC of venlafaxine increased by 21% in X-Met and by 70% in P-Met. EFA AUC values increased by 23% and 33% in X-Met and P-Met subjects, respectively. Drugs that affect blood clotting and platelet function (NSAIDs, acetylsalicylic acid preparations and other anticoagulants) Serotonin, released by platelets, plays an important role in hemostasis (stopping bleeding). Epidemiological studies demonstrate an association between the use of psychotropic medications that interfere with serotonin reuptake and the incidence of upper gastrointestinal bleeding. This relationship is enhanced if NSAIDs, drugs containing acetylsalicylic acid or other anticoagulants are used simultaneously. The risk of bleeding has been shown to increase when SSRIs and SNRIs (including venlafaxine) are prescribed concomitantly with warfarin. Patients prescribed warfarin should be closely monitored for prothrombin time and/or partial thromboplastin time, especially when co-administration with venlafaxine is started or stopped. Interaction with other drugs at the level of the studied metabolism with cytochrome P450 isoenzymes The main routes of metabolism of venlafaxine include isoenzymes CYP2D6 and CYP3A4: the first of them converts venlafaxine into its active metabolite EDV, and the second is less important in the metabolism of venlafaxine compared to CYP2D6 and forms the product N -desmethylvenlafaxine with little pharmacological activity. Preclinical studies have shown, and subsequently confirmed clinically, that venlafaxine is a relatively weak inhibitor of CYP2D6. Therefore, even when prescribed with drugs that moderately suppress the activity of this enzyme (see the example with imipramine above), or in the case of patients with a genetically determined decrease in CYP2D6 function, no dose adjustment of venlafaxine is required, since the total concentration of the active substance and active metabolite (venlafaxine and EFA) does not change significantly. This characterizes venlafaxine positively when compared with other antidepressants. Caution should be exercised when co-administered with CYP2D6 inhibitors such as quinidine, paroxetine, fluoxetine, haloperidol, perphenazine, levomepromazine, as venlafaxine may potentially increase the plasma concentrations of these CYP2D6 substrates. In combination with drugs that inhibit both enzymes (CYP2D6 and CYP3A4), special caution is required. Such drug interactions have not yet been sufficiently studied, and in this case this combination of drugs is not recommended. In addition, venlafaxine does not inhibit the activity of the enzymes CYP3A4, CYP1A2 and CYP2C9, therefore, no significant interaction is observed with drugs such as alprozolam, caffeine, carbamazepine, diazepam, tolbutamide, terfenadine. Interaction with ketoconazole is described above. CYP3A3/4 inhibitors such as itraconazole and ritonavir can have a similar effect. Other interactions with various concomitant therapeutic factors and food When using venlafaxine, special caution should be used during electroconvulsive therapy, as there is no experience with the use of venlafaxine in these conditions. There was no significant effect of different types of food on the absorption of venlafaxine and its subsequent conversion to EDV. Foods (usually high in protein, for example: hard cheeses, fish roe, turkey), as well as nutritional supplements and fitness diets, which are an increased source of tryptophan, potentially contribute to greater production of serotonin in the body, which may increase serotonergic side effects. effects of venlafaxine. Undesirable pharmacodynamic interactions may occur when Venlafaxine is taken simultaneously with the medicinal plant St. John's wort (herb or various preparations made from it); such a combination is not recommended. There are reports of false-positive results of the immunochromatographic rapid urine test (test strip) for phencyclidine and amphetamines in patients taking venlafaxine, even several days after discontinuation of venlafaxine. This may be explained by the lack of specificity of this test. Only a confirmatory test in a specialized anti-doping laboratory can distinguish venlafaxine from phencyclidine and amphetamines. According to the data available to date, venlafaxine has not shown itself to be a drug that causes drug abuse or addiction (both in preclinical receptor affinity studies and in clinical practice).
Side effect
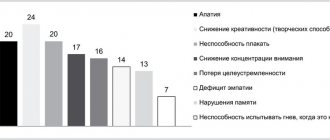
Frequency of side effects: very often (? 1/10), often (? 1/100 to General symptoms: - often - weakness, fatigue, chills; - infrequently - Quincke's edema, photosensitivity reactions; - frequency not established - anaphylactic reactions. From the nervous system: - very often - dry mouth, headache; - often - unusual dreams, decreased libido, dizziness, insomnia, increased excitability, paresthesia, stupor, confusion, depersonalization, increased muscle tone, tremor; - infrequently - apathy, agitation, hallucinations, myoclonus, impaired coordination of movements and balance; - rarely - akathisia, psychomotor agitation, epileptic seizures, manic reactions; - frequency not established - dizziness, neuroleptic malignant syndrome (NMS), serotonin syndrome, delirium, extrapyramidal reactions ( including dystonia and dyskinesia), tardive dyskinesia, suicidal thoughts and behavior, aggression. From the gastrointestinal tract: - very often - nausea; - often - loss of appetite (anorexia), constipation, vomiting; infrequently - bruxism, diarrhea; - rarely - hepatitis; - frequency not established - pancreatitis. From the respiratory system: - often - yawning, bronchitis, shortness of breath; - rarely - interstitial lung diseases and eosinophilic pneumonia, chest pain. From the cardiovascular system: - often - arterial hypertension, hyperemia of the skin; - uncommon - postural hypotension, tachycardia, fainting; - frequency not established - hypotension, QT interval prolongation, ventricular fibrillation, ventricular tachycardia (including bidirectional tachycardia). From the hematopoietic system: - infrequently - hemorrhages into the skin (ecchymosis), gastrointestinal bleeding; - frequency not established - hemorrhages in the mucous membranes, prolongation of bleeding time, thrombocytopenia, pathological changes in the blood (including agranulocytosis, aplastic anemia, neutropenia and pancytopenia). From the metabolic side: - often - increased cholesterol levels in the blood serum, decreased body weight; - uncommon - weight gain; - very rarely - increase in prolactin levels; frequency not established - changes in laboratory tests of liver function, hepatitis, hyponatremia, syndrome of insufficient secretion of antidiuretic hormone. From the genitourinary system: - often - ejaculation/orgasm disorders (in men), erectile dysfunction (impotence), anorgasmia, dysuric disorders (mainly difficulties at the beginning of urination), pollakiuria, menstrual disorders associated with increased bleeding or increased irregular bleeding (menorrhagia, metrorrhagia); - infrequently - orgasm disturbances (in women), urinary retention; rarely - urinary incontinence. From the senses: - often - disturbances of accommodation, mydriasis, blurred vision; - infrequently - disturbance of taste, noise or ringing in the ears; — frequency not established — angle-closure glaucoma. From the skin: - very often - sweating; - infrequently - alopecia, rapidly passing rash; - frequency not established - erythema multiforme, toxic epidermal necrolysis, Stevens-Johnson syndrome, itching, urticaria. From the musculoskeletal system: frequency not established - rhabdomyolysis. When you stop taking venlafaxine, abruptly stop or reduce the dose, symptoms may be observed that relate to the so-called withdrawal syndrome: increased fatigue, asthenia, headache, dizziness, sleep disturbances (drowsiness or insomnia, difficulty falling asleep, the appearance of unusual dreams), hypomania, anxiety, agitation (increased nervous excitability and irritability), confusion, paresthesia (spontaneously occurring unpleasant sensation of numbness, tingling, burning, crawling, etc.), increased sweating, dry mouth, decreased appetite, nausea, vomiting, diarrhea (most of these reactions are mild and do not require treatment).
Contraindications
- hypersensitivity to venlafaxine or any of the excipients;
- simultaneous use with MAO inhibitors; - severe renal dysfunction (glomerular filtration rate (GFR) less than 10 ml/min); - severe liver dysfunction; - taking the drug under the age of 18 years; - during pregnancy and lactation. With caution: recent myocardial infarction, unstable angina, arterial hypertension, arrhythmias (especially tachycardia), history of convulsive syndrome, increased intraocular pressure, closed-angle glaucoma, history of manic states, suicidal tendencies, predisposition to bleeding from the skin and mucous membranes , initially reduced body weight, hyponatremia, dehydration, simultaneously with diuretics or drugs used to treat obesity. Use during pregnancy and breastfeeding Venlafaxine should not be prescribed to pregnant and breastfeeding women, because The safety of the drug during pregnancy and lactation in women has not been sufficiently established, due to the fact that there are no adequately controlled clinical studies on a sufficiently large sample of such patients. This concerns the health of both the mother and, to a greater extent, the fetus/child. Women of childbearing age should be warned about this before starting treatment and should immediately consult a doctor if they become pregnant or plan to become pregnant during treatment with the drug. Venlafaxine and its metabolite (EFV) are excreted into breast milk. If it is necessary to take the drug during lactation, breastfeeding must be stopped. In practice, there are cases of prescribing venlafaxine to mothers during pregnancy and shortly before childbirth, when in a particular situation the expected benefit to the mother outweighs the potential risk to the fetus. In these cases, newborns often experienced complications that required: increased hospitalization, maintenance of breathing and tube feeding. These complications can develop immediately after childbirth and are also typical when taking other antidepressants from the SNRI group or SSRIs (not containing venlafaxine). In such cases, the following clinical symptoms in newborns have been reported: respiratory disorders, cyanosis, apnea, convulsions, temperature instability, feeding difficulties, vomiting, hypoglycemia, muscle hypertension or hypotonia, hyperreflexia, tremor, trembling, irritability, lethargy, constant crying, drowsiness or insomnia. Such disturbances may indicate the serotonergic effects of the drug Venlafaxine. If venlafaxine was used during pregnancy and maternal treatment was completed shortly before delivery, the newborn may experience withdrawal symptoms. In such a newborn, the presence of serotonin syndrome or neuroleptic malignant syndrome should be excluded. Epidemiological evidence suggests that use of SSRIs during pregnancy, especially late in pregnancy, may increase the risk of persistent neonatal pulmonary hypertension.
Overdose
Symptoms of overdose: impaired consciousness (from drowsiness to coma), agitation, possible vomiting, diarrhea; tremor, decrease or (mild) increase in blood pressure, dizziness, mydriasis, convulsive states, sinus or ventricular tachycardia or bradycardia; changes on the ECG (prolongation of the QT interval, bundle branch block, widening of the QRS complex). Post-marketing experience indicates that the most common overdose of venlafaxine occurred with concomitant use of alcohol and/or other psychotropic drugs. There are repeated reports of deaths. Published literature on retrospective studies of venlafaxine overdoses report that this increased risk of fatal outcomes may be inherent in venlafaxine when compared with commercially available SSRI antidepressants, but this risk is lower than the risk inherent in tricyclic antidepressants. Epidemiological studies have shown that those patients treated with venlafaxine have a greater risk of suicide compared with those patients treated with SSRIs (other than venlafaxine). However, it remains unclear to what extent these high rates of death (due to venlafaxine overdose) are due to the toxic properties of the drug itself or the special characteristics of the group of patients treated with venlafaxine. According to clinical experience, it is recommended that prescriptions for venlafaxine prescribe the minimum possible amount of the drug, sufficient only until the patient's next visit, in order to reduce the risk of intentional overdose. Treatment: symptomatic and supportive therapy is provided. Specific antidotes are unknown. Continuous monitoring of vital functions (respiration, circulation and heart rate) is recommended. In case of overdose, it is recommended to immediately lavage the stomach and take activated charcoal to reduce the absorption of the drug. Inducing vomiting is not recommended if there is a risk of aspiration of vomit. Forced diuresis, dialysis, and blood transfusion are ineffective.
special instructions
Suicide and suicidal behavior Depression is associated with an increased risk of suicidal ideation, self-harm, and suicide (suicidal behavior). This risk persists until significant remission occurs. Since improvement may not be observed during the first few weeks of therapy or even longer, patients should be closely monitored until such improvement occurs. Based on accumulated clinical experience, the risk of suicide may increase in the early stages of recovery. Patients with a history of suicide attempts or high levels of suicidal ideation prior to treatment are at greater risk for suicidal ideation or suicide attempts and should be closely monitored. A meta-analysis of placebo-controlled clinical trials of antidepressants in adult patients with mental disorders found that antidepressants were at increased risk of suicidal behavior compared with placebo in patients under 25 years of age. Drug treatment of these patients, and in particular those at high risk of suicide, should be accompanied by careful monitoring, especially early in therapy and during dose adjustment. Patients (and caregivers of such patients) should be warned to monitor for any signs of clinical worsening, suicidal behavior or ideation, or unusual changes in behavior, and to seek immediate medical attention if these symptoms occur. In a small number of patients taking antidepressants, incl. venlafaxine, aggression may occur during initiation of treatment, dose changes, or discontinuation of treatment. Clinical studies conducted to date have not revealed tolerance or dependence to venlafaxine. Despite this, as with other drugs that act on the central nervous system, the physician should closely monitor patients for signs of drug abuse, as well as patients with a history of such symptoms. Special Populations Venlafaxine is not approved for use in children. In patients with a history of aggression, venlafaxine should be used with caution. In patients with affective disorders, bipolar disorder when treated with antidepressants, incl. venlafaxine, hypomanic and manic states may occur. Like other antidepressants, venlafaxine should be used with caution in patients with a history of mania. Such patients require medical supervision. Convulsive disorders may occur during therapy with venlafaxine. As with all antidepressants, venlafaxine should be used with caution in patients with a history of seizure disorders and such patients should be closely monitored. Treatment should be discontinued if seizures develop. Akathisia The use of venlafaxine has been associated with the development of akathisia, which is characterized by an unpleasant feeling of internal motor restlessness for the patient and manifested in the patient’s inability to sit quietly in one position for a long time or remain motionless for a long time. This condition may occur at the beginning of treatment and during the first weeks of treatment. In patients who develop such symptoms, increasing the dose is not recommended. Bipolar disorder Before initiating treatment, it is necessary to identify those patients who are at risk for bipolar disorder. Such a check should include a detailed examination of the medical history, incl. family, to identify cases of suicide, bipolar disorder. It should be noted that venlafaxine is not recommended for use in the treatment of bipolar depression. Use in patients with concomitant diseases Clinical experience with the use of venlafaxine in patients with concomitant diseases is limited. It should be used with caution in patients with diseases in which the effect of venlafaxine on hemodynamic parameters and/or metabolism may be significant. Patients should be warned to immediately consult a doctor if a rash, urticaria or other allergic reactions occur. Some patients while taking venlafaxine experienced a dose-dependent increase in blood pressure and/or an increase in heart rate, so regular monitoring of blood pressure and ECG is recommended, especially during the period of adjusting or increasing the dosage of venlafaxine. In post-marketing experience with venlafaxine (overdose), fatal cardiac arrhythmias have been reported. Before prescribing venlafaxine to patients at high risk of developing serious cardiac arrhythmias, the ratio of the likely benefit to the possible risk of use should be assessed. Patients, especially the elderly, should be warned about the possibility of dizziness and impaired balance in order to prevent injury. While taking venlafaxine, especially in conditions of dehydration or decreased blood volume (including in elderly patients and patients taking diuretics), hyponatremia and/or syndrome of insufficient secretion of antidiuretic hormone may occur. Venlafaxine has not been studied in patients with recent myocardial infarction and decompensated heart failure. The drug should be prescribed to such patients with caution. Taking SSRIs or venlafaxine in patients with diabetes may cause changes in plasma glucose levels. Dosage adjustments of insulin and/or antidiabetic medications may be required. During treatment, it is recommended to refrain from drinking any alcohol-containing drinks. The safety and effectiveness of venlafaxine in combination with weight loss medications (including phentermine) have not been established. The simultaneous use of venlafaxine and drugs that reduce body weight is not recommended. Women of childbearing potential should use appropriate contraception while taking venlafaxine. Explanations of special symptoms and conditions that may occur during treatment with the drug Dry mouth is observed in 10% of patients receiving venlafaxine. This may increase your risk of developing tooth decay. Patients should practice good oral hygiene. The use of venlafaxine may cause the development of akathisia, characterized by subjective discomfort or restlessness and the need to move frequently, often accompanied by an inability to sit or stand still. This mostly occurs during the first few weeks of treatment. Increasing the dose in patients who develop these symptoms may cause undesirable effects. In placebo-controlled clinical trials, a clinically significant increase in serum cholesterol was reported in 5.3% of patients. Cholesterol level control is necessary during long-term treatment. Withdrawal syndrome When stopping treatment, withdrawal syndrome is common, especially if it is stopped abruptly. The risk of withdrawal syndrome may depend on several factors, including the duration of treatment, the size of therapeutic doses and the rate of dose reduction. These symptoms are very rarely reported in patients who accidentally missed taking the drug. Withdrawal symptoms usually occur within the first few days after stopping treatment. These symptoms usually go away within 2 weeks, although in some people they may last 2-3 months or more. It is recommended to gradually reduce the dose of venlafaxine when stopping the drug - over several weeks or months, depending on the severity of the clinical symptoms of the disease. Serotonin syndrome Venlafaxine, like other serotonergic drugs, can cause serotonin syndrome, a potentially life-threatening condition, especially when co-administered with other drugs that can affect serotonergic neurotransmitter systems such as MAO inhibitors. Symptoms of serotonin syndrome may include mental status changes (agitation, hallucinations, coma), autonomic instability (tachycardia, blood pressure lability, hyperthermia), neuromuscular disorders (hyperreflexia, incoordination), and/or gastrointestinal symptoms (nausea, vomiting). , diarrhea). Effect on the ability to drive vehicles and operate machinery During the treatment period, care should be taken when performing potentially hazardous types of work that require increased concentration and psychomotor reaction speed (including driving a car and operating machinery).
Dispensing conditions in pharmacies
On prescription