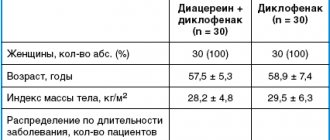
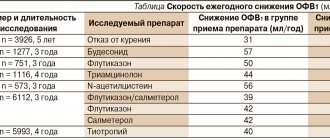
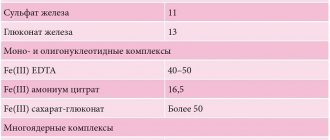
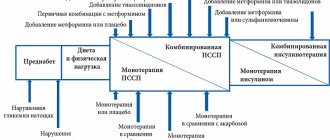
Interaction
With the simultaneous use of the drug Xefocam® and
cimetidine - increased plasma concentrations of lornoxicam;
ranitidine and antacid drugs - no interaction detected;
anticoagulants or platelet aggregation inhibitors - possible increase in bleeding time (increased risk of bleeding, MHO control is required);
β-blockers and ACE inhibitors - their hypotensive effect may be reduced;
diuretics - decreased diuretic effect and hypotensive effect;
digoxin - decreased renal clearance of digoxin;
quinolone antibiotics - increased risk of developing seizures;
other NSAIDs or corticosteroids - increased risk of gastrointestinal bleeding;
methotrexate - increased concentration of methotrexate in serum;
SSRIs (for example, citalopram, fluoxetine, paroxetine, sertraline) - increased risk of gastrointestinal bleeding;
lithium salts - it is possible to increase peak plasma lithium concentrations and thereby increase the known side effects of lithium;
cyclosporine - increased nephrotoxicity of cyclosporine;
sulfonylurea derivatives - enhancing the hypoglycemic effect of the latter;
alcohol, corticotropin, potassium supplements - increased risk of side effects from the gastrointestinal tract;
cefamandole, cefoperazone, cefotetan, valproic acid - increased risk of bleeding.
Contraindications
known hypersensitivity/allergy to lornoxicam or to one of the components of the drug;
complete or incomplete combination of bronchial asthma, recurrent nasal polyposis or paranasal sinuses and intolerance to acetylsalicylic acid and other NSAIDs (including a history);
bleeding diathesis or bleeding disorders, as well as patients who have undergone operations associated with the risk of bleeding or incomplete hemostasis;
period after coronary artery bypass surgery;
erosive and ulcerative changes in the mucous membrane of the stomach or duodenum, active gastrointestinal bleeding;
cerebrovascular or other bleeding;
recurrent stomach ulcer or repeated gastrointestinal bleeding;
history of gastrointestinal bleeding associated with taking NSAIDs;
inflammatory bowel diseases (Crohn's disease, ulcerative colitis) in the acute phase;
decompensated heart failure;
severe liver failure or active liver disease;
severe renal failure (serum creatinine level more than 300 µmol/l), progressive kidney disease;
confirmed hyperkalemia, hypovolemia or dehydration;
pregnancy, breastfeeding period;
children under 18 years of age (due to the lack of clinical data on its use in this age group).
With caution: erosive and ulcerative lesions and bleeding from the gastrointestinal tract (history); moderate renal failure; conditions after surgical interventions; age over 65 years; IHD, chronic heart failure; cerebrovascular diseases; dyslipidemia/hyperlipidemia; diabetes; peripheral arterial disease; smoking; Cl creatinine less than 60 ml/min; history of ulcerative lesions of the gastrointestinal tract; presence of Helicobacter pylori infection; long-term use of NSAIDs; alcoholism; severe somatic diseases; simultaneous use of oral corticosteroids (including prednisolone), anticoagulants (including warfarin), antiplatelet agents (including clopidogrel), SSRIs (including citalopram, fluoxetine, paroxetine, sertraline).
Pharmacodynamics
It has a pronounced analgesic and anti-inflammatory effect. Lornoxicam has a complex mechanism of action, which is based on the suppression of PG synthesis due to inhibition of the activity of COX isoenzymes. In addition, lornoxicam inhibits the release of oxygen free radicals from activated leukocytes.
The analgesic effect of lornoxicam is not associated with narcotic effects.
The drug Xefocam® does not have an opiate-like effect on the central nervous system and, unlike narcotic analgesics, does not depress respiration and does not cause drug dependence.
Manufacturer
Nycomed Denmark ApS, Denmark. Helseholmen 9, DK-2650, Hvidovr, Denmark.
Lenitzstrasse 70–98, D-16515, Oranienburg, Germany.
Filling/packaging, releasing quality control. Nycomed Austria GmbH, Austria. Art. Peterstrasse 25, A-4020, Linz, Austria.
Nycomed Denmark ApS, Denmark. Langebjerg 1, DK-4000, Roskilde, Denmark.
Lenitzstrasse 70–98, D-16515, Oranienburg, Germany.
Consumer complaints should be sent to Takeda Pharmaceuticals LLC. 119048, Moscow, st. Usacheva, 2, building 1.
Tel.; Fax.
www.takeda.com;
special instructions
The risk of the ulcerogenic effect of the drug can be reduced by the simultaneous administration of proton pump inhibitors and synthetic analogues of PG. If bleeding occurs in the gastrointestinal tract, the drug should be stopped immediately and appropriate emergency measures taken. It is especially necessary to carefully monitor the condition of those patients with gastrointestinal pathology who are receiving a course of treatment with Xefocam® for the first time.
Like other oxicams, it inhibits platelet aggregation and may therefore increase bleeding time. When using this drug, it is necessary to carefully monitor the condition of patients who require absolutely normal functioning of the blood coagulation system (for example, patients who are undergoing surgery), who have disorders of the blood coagulation system, or who are receiving drugs that inhibit coagulation (including low-dose heparin), for in order to promptly detect signs of bleeding.
If signs of liver damage appear (itching, yellowing of the skin, nausea, vomiting, abdominal pain, dark urine, increased levels of liver transaminases), you should stop taking the drug and consult your doctor.
The drug should not be used simultaneously with other NSAIDs.
The drug can change the properties of platelets, but does not replace the preventive effect of acetylsalicylic acid in cardiovascular diseases.
For patients with impaired renal function caused by large blood loss or severe dehydration, the drug Xefocam®, as an inhibitor of PG synthesis, can be prescribed only after hypovolemia and the associated risk of reducing renal perfusion have been eliminated. Like other NSAIDs, it can cause an increase in blood urea and creatinine concentrations, as well as water and sodium retention, peripheral edema, hypertension and other early signs of nephropathy. Long-term treatment of such patients with Xefocam® can lead to the following consequences: glomerulonephritis, papillary necrosis and nephrotic syndrome with transition to acute renal failure. The drug should not be prescribed to patients with a pronounced decrease in renal function (see “Contraindications”). In elderly patients, as well as in patients suffering from arterial hypertension and/or obesity, it is necessary to control blood pressure levels.
It is especially important to monitor renal function in elderly patients, as well as in patients simultaneously receiving diuretics and drugs that can cause kidney damage.
With long-term use of the drug Xefocam®, it is necessary to periodically monitor hematological parameters, as well as renal and liver function. The use of the drug may adversely affect female fertility and is not recommended for women planning pregnancy.
Patients using the drug must refrain from activities that require increased attention, rapid mental and motor reactions, and alcohol consumption.
Side effects
From the gastrointestinal tract and liver: dyspepsia, abdominal pain, dry mouth, stomatitis, nausea, vomiting, heartburn, diarrhea, esophagitis, gastritis, erosive and ulcerative lesions of the mucous membrane of the stomach and intestines, incl. with perforation and bleeding, constipation, flatulence, melena, liver dysfunction, increased levels of liver transaminases.
From the nervous system: headache, dizziness, drowsiness, sleep disturbances, depression, agitation, tremor, aseptic meningitis, paresthesia.
From the skin and subcutaneous fat: edematous syndrome, ecchymosis, skin rash, itching, urticaria, alopecia, Stevens-Johnson syndrome, Lyell's syndrome, angioedema.
From the urinary system: dysuria, decreased glomerular filtration, interstitial nephritis, glomerulonephritis, papillary necrosis, nephrotic syndrome, peripheral edema, acute renal failure.
From the senses: tinnitus, blurred vision.
From the cardiovascular system: development or worsening of heart failure, tachycardia, increased blood pressure.
From the hematopoietic organs and hemostasis system: agranulocytosis, leukopenia, anemia, thrombocytopenia, increased bleeding time.
From the respiratory system: pharyngitis, rhinitis, dyspnea, cough, bronchospasm.
Other: anorexia, increased sweating, changes in body weight, arthralgia, myalgia.
Pharmacokinetics
Lornoxicam is quickly and almost completely absorbed from the gastrointestinal tract after oral administration. In this case, Cmax in plasma is achieved in approximately 1–2 hours. Food intake reduces Cmax by 30% and increases Tmax up to 2.3 hours. The absolute bioavailability of lornoxicam is 90–100%. Lornoxicam is present in plasma mainly unchanged and, to a lesser extent, in the form of a hydroxylated metabolite, which has no pharmacological activity. The binding of lornoxicam to plasma proteins (mainly to the albumin fraction) is 99% and does not depend on its concentration.
T1/2, on average, is 4 hours and does not depend on the concentration of the drug. Lornoxicam is completely metabolized in the liver. The isoenzyme CYP2C9 is involved in metabolism. Approximately 1/3 of the metabolites are excreted from the body by the kidneys and 2/3 by bile. In elderly people, as well as in patients with renal or hepatic insufficiency, no significant changes in the pharmacokinetics of lornoxicam were found.
Directions for use and doses
Inside.
Take before meals with a glass of water.
For severe pain: the recommended dose is 8–16 mg/day, divided into 2–3 doses; the maximum daily dose is 16 mg.
For inflammatory and degenerative rheumatic diseases: the recommended initial dose is 12 mg; the standard dose is 8–16 mg/day, depending on the patient’s condition.
The duration of therapy depends on the nature and course of the disease.
For diseases of the gastrointestinal tract, patients with impaired renal or liver function, elderly people (over 65 years old), after major operations, a maximum daily dose of 12 mg is recommended, divided into 3 doses during the day.
To reduce the risk of developing adverse events from the gastrointestinal tract, the minimum effective dose should be used for the shortest possible short course.