pharmachologic effect
Human anti-rhesus immunoglobulin RhO(D) is a protein fraction with immunological activity. It is isolated from the serum or plasma of those donors who were initially tested for the absence of antibodies to HIV , hepatitis C in the blood, and the surface antigen of the hepatitis B virus was also absent in the blood. The active ingredient of the drug is immunoglobulin G, which contains incomplete anti-Rho ( D) antibodies. Under the influence of this drug in the body of a pregnant woman with negative Rh, who has given birth to children with positive Rho (D), or who has undergone an abortion if the man's blood is Rho (D) positive, Rh sensitization is prevented (that is, the formation of Rho (D) antibodies).
Detailed description of the study
Immunoglobulins (Ig, antibodies) are serum proteins that are factors of humoral immunity. This subtype of the immune system protects the human body from various extracellular microorganisms (mainly bacteria, helminths) and their toxins.
Immunoglobulins have a general structural plan, but differ from each other in some structural areas. Based on this, five classes of immunoglobulins are distinguished: M (IgM), G (IgG), A (IgA), E (IgE) and D (IgD). This laboratory test is aimed at determining the concentration of immunoglobulins IgA, IgM and IgG in the blood.
IgA immunoglobulins are predominantly present in the secretions of glandular organs: saliva, digestive juice, secretions of the nasal mucosa and mammary gland. In the blood, the content of these antibodies is limited to 10-15% of the total amount of all immunoglobulins.
One of the key functions of class A immunoglobulins is the body's first line of defense on mucosal surfaces. IgA prevents the penetration of various microorganisms and their waste products. Although IgA does not have bactericidal activity, it plays an important role in neutralizing bacterial toxins. It is also known that immunoglobulins A are widely represented in colostrum (breast milk) and can provide specific immune protection for the newborn against various infections.
Class M immunoglobulins have a complex structure and the highest molecular weight. In this regard, IgM antibodies cannot be transmitted (through the placental barrier) from the mother’s body to the fetus during pregnancy.
IgM perform one of the leading functions in immune defense; they are the earliest antibodies that are produced in response to foreign molecules (antigens). Immunoglobulins M can participate in the neutralization of antigens independently, as well as as part of a complex immune complex - the complement system. The greatest activity of IgM is manifested against many bacteria - pathogens of pneumonia, meningitis, Haemophilus influenzae and others.
IgG antibodies are the dominant class in terms of the number of molecules in blood serum - they account for 75-80% of all immunoglobulins. IgG is predominantly produced after IgM antibodies during the secondary immune response (when a foreign molecule enters the body again). This property determines one of the features of IgG - these antibodies contribute to the long-term immune protection of the body from various bacteria, viruses, helminths, protozoa and their metabolic products.
Due to their small size, IgG are the only immunoglobulins that can cross the placental barrier during pregnancy. They are considered the main antibodies that provide immunity to a newborn baby during the first 3-6 months of his life.
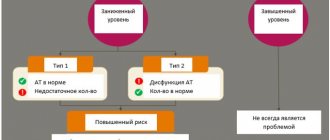
The main pathologies with immunoglobulins of classes A, M and G are associated with a violation of their quantity. The lack of these antibodies is often associated with immunodeficiency, which can have various origins. Deficiency of IgA, IgM and IgG may be accompanied by the appearance or exacerbation of infectious diseases (respiratory tract, ENT organs, etc.); autoimmune (rheumatoid arthritis, systemic lupus erythematosus, etc.) and allergic pathologies (rhinitis, asthma, etc.).
The causes of excess antibodies of the IgA, IgM and IgG classes are most often acute or recurrent inflammatory diseases (infectious and non-infectious nature), as well as oncohematological pathologies associated with tumor degeneration of B-lymphocytes and uncontrolled formation of immunoglobulins of one or more classes.
This laboratory test is recommended for patients to assess the humoral part of the immune system as part of the diagnosis of (acute/chronic) infectious, autoimmune, hematological and oncological diseases, as well as immunodeficiency states. In the Hemotest laboratory you can also additionally undergo a comprehensive study of humoral immunity.
A detailed description of the studies and reference values are presented on the pages with descriptions of individual studies.
Indications for use
This product can only be used as directed by a specialist. The drug is indicated for use to ensure the prevention of Rh conflict in women who have a negative Rh factor in the following cases:
- during pregnancy and subsequent birth of a child with a positive Rh factor;
- in case of spontaneous or intentional termination of pregnancy ;
- in case of termination of an ectopic pregnancy ;
- if there is a threat of spontaneous abortion in any trimester;
- after amniocentesis , as well as other procedures in which there is a risk that fetal blood may be in the mother’s blood;
- in case of abdominal trauma.
Total immunoglobulin D (IgD)
Our goal is to make the test procedure convenient and accessible so that you can take care of your health!
All studies are performed in the ArchiMed research medical laboratory - one of the leading medical laboratories in Europe!
So, why should you choose us?
IT COULD NOT BE EASIER
- Add the necessary tests to your cart.
- Select the date for the tests and make payment.
- Submit the biomaterial for research.
You receive test results in your personal account and by email when they are ready.
Having difficulty finding the necessary tests? Entrust it to us! Provide a list of assigned studies to our operators in an online chat with a request to create a cart. All you have to do is check and place your order!
SAVE UP TO 50%
By placing an order on the website, you save up to 50%!
Why? Because we do not open or maintain test collection points, as other laboratories do. We have eliminated all additional costs for cashiers, administrators, rent, etc., directing the money to use modern equipment and reagents from the world's best manufacturers! And you can use the saved budget for additional regular research or other important everyday expenses.
RELIABLE PROTECTION
We do not use cloud storage!
Our software package uses dedicated communication channels and the laboratory’s own servers. Therefore, all information we receive is under reliable protection, and its collection and processing is carried out in compliance with the requirements for the protection of personal data.
THE MOST ADVANCED RESEARCH TECHNOLOGIES
Today we have the most high-precision automated equipment
, and the modern technologies and highly qualified specialists we use allow us to achieve unsurpassed results in the diagnostic process. This allows us not only to carry out research in the shortest possible time, but also to achieve the most accurate results! We never rest on our laurels and therefore develop and implement new methods in such promising areas of diagnostics as molecular genetics and mass spectrometry.
ONLY IMPORTED REAGENTS For diagnostics we use exclusively imported reagents and reagents
, as well as the best containers in their segment for transporting biomaterial to the laboratory in its unchanged form. This also allows us to achieve the best results and perform ultra-accurate diagnostics!
ANALYSIS-MARKET IS YOUR RELIABLE ASSISTANT! Our team works 7 days a week and does everything to make the test procedure simple, convenient, accessible and understandable for you!
Side effects
Adverse reactions are rare. When this drug is administered, the following manifestations are possible:
- hyperemia of the skin in the places where the solution is injected;
- on the first day after the injection - increased body temperature, dyspeptic symptoms;
- various allergic manifestations .
, anaphylactic shock may develop . Patients who have been administered the medicine must remain under the supervision of specialists for half an hour after the injection. The specialist should have the opportunity to carry out anti-shock treatment if necessary.
The beginning of the use of drugs from human blood for the prevention and treatment of infectious diseases dates back to 1910, when researchers from the Pasteur Institute in France M. Nicolle and E. Consei proposed using convalescent blood serum to prevent typhus in children [1], and in 1918 M. Nicolle and E. Consei, as well as DL Richardson and H. Connor, independently of each other, successfully used the serum of children who recovered from measles to prevent this disease [2]. In the USSR, this area of prevention was started in 1925 [3]. Homologous sera had prophylactic effectiveness against a number of infectious diseases, but there were many problems with their production and use. The cost of the resulting serum was high, and at the same time, patients required large doses of the drug. For example, to prevent measles, it was necessary to administer from 15 to 40 ml of serum to children under 2 years of age; the procedure was painful and unsafe due to the high probability of developing abscesses at the injection site [4]. Practical healthcare required cheaper, safer and more effective immunobiological drugs.
In 1946, a group of researchers led by H. Gold and JE Cohn developed a unique technology for separating plasma fractions, which later became classical, and characterized each of the fractions. It was found that the protein fraction, called gamma globulin, consists of a mass of antibodies against pathogens of various diseases. The first commercial drug in the USA was released in a 2 ml bottle under the name “immune serum globulin (human)”. It was an approximately 16% solution containing the antibodies present in the normal plasma pool, but at a concentration almost 25 times greater. Antibodies to the causative agents of measles, scarlet fever, mumps, whooping cough, rubella, chicken pox, neonatal diarrhea, polio, serum hepatitis, infectious hepatitis, and influenza were identified in the drug. According to research results, the drug showed the highest effectiveness when used for the prevention of measles and infectious hepatitis [5].
In the USSR in 1948, researchers at the Moscow Institute of Epidemiology and Bacteriology N.V. Kholchev and L.I. Kolesnikov’s method for obtaining gamma globulin from serum, developed by Kohn and co-workers, was modified; instead of centrifuging the sediment, a filtration method was proposed [6]. The technique was adopted for use in the production of gamma globulin for the prevention of measles [7]. To prevent measles, a drug containing at least 18% protein was administered intramuscularly in a volume of 3.0 ml (corresponding to 60 ml of anti-measles serum) [8].
In 1952, military doctor O. Bruton (USA) successfully used the fraction of human gamma globulin obtained by Cohn and co-workers [9] in the form of an intramuscular injection for the prevention of chronic lung diseases and other bacterial infections in children with extremely low levels of immunoglobulins, identified by electrophoresis of blood samples [10]. In 1952–1953 gamma globulin was included in US medical standards [11]. Thus, the beginning of the widespread use of normal human immunoglobulin (HNI) preparations was laid in the replacement therapy of primary and some secondary immunodeficiencies (absence or decreased level of antibody production), as well as immunomodulatory therapy. In the middle of the twentieth century. gamma globulin was actively used for the prevention of measles, rubella, chickenpox, polio, mumps and infectious hepatitis [12–15].
Since the experience of research and practical application has shown the effectiveness of gamma globulin in the treatment and prevention of various diseases, at a seminar on gamma globulin held in Paris in December 1954, it was decided to change the international name “measles gamma globulin” to the more general one - "gamma globulin". In 1966, the expert committee on the use of human immunoglobulin at the World Health Organization (WHO) recommended the use of a single international name for the drug “immunoglobulin” [16].
The effectiveness and safety of the medical use of HCI drugs are determined by the fact that the antibodies they contain can specifically interact with foreign antigens, as well as the ability of the drug to cause nonspecific effects [17]. Before 1981 (before the advent of deep purification methods), immunoglobulin preparations were almost always used intramuscularly. Disadvantages that cannot be resolved by the technology for producing immunoglobulin preparations for intramuscular administration are pain at the injection site, low rate of entry of antibodies into the systemic circulation and the inability to quickly create high concentrations of antibodies in situations requiring urgent therapeutic measures. Various side effects that develop in response to the administration of such drugs are collectively called “serum sickness” - joint pain, fever, swollen lymph nodes and erythema [4]. With intravenous administration of immunoglobulins for intramuscular administration, patients may develop anaphylactoid reactions associated with nonspecific activation of complement as a result of spontaneous formation of immunoglobulin aggregates and the presence of trace amounts of proteases in the drug [18].
Less toxic subcutaneous administration of HCI drugs was practiced less frequently, because this method of drug administration was limited in volume (usually no more than 5 ml per injection), which did not allow its use as a means for immunoreplacement therapy [19].
The report of the expert committee on the use of immunoglobulin at the WHO [16] noted that since 1945, when gamma globulin began to be used for the prevention of measles and infectious hepatitis, the scope of its use has expanded significantly and developed in two directions: the prevention of certain viral diseases (measles, infectious hepatitis and rubella); treatment of antibody deficiency syndromes to prevent recurrent bacterial infections. However, the report stated that the regulatory framework governing the use of gamma globulin should be reviewed because The development and widespread use of safe and effective vaccines reduces the need for passive immunization and, accordingly, the use of gamma globulin.
The authors of the report, based on the results of numerous clinical data (including with the participation of military contingents in various countries), recognized that HCI is not effective for the prevention and treatment of influenza and other respiratory viral infections.
According to modern WHO requirements and experience in the practical use of drugs, intramuscular administration of HCI is limited to a short list of indications: prevention of hepatitis A, as well as some specific infections (for example, measles) [20, 21]. Such drugs have been shown to be effective in preventing bacterial infections in children with hereditary agammaglobulinemia [22].
In the Russian Federation, 8 HCI drugs for intramuscular administration are registered [23].
The instructions for the use of domestic drugs indicate the following indications: prevention of hepatitis A, prevention of measles, prevention and treatment of influenza, prevention of whooping cough, prevention of meningococcal infection, prevention of poliomyelitis, treatment of hypo- and agammaglobulinemia in children, increasing the body's resistance during the period of convalescence of acute infectious diseases with protracted course and chronic pneumonia. At the same time, the list of indications in the instructions for use of various drugs varies significantly and requires revision. The results of numerous clinical studies have confirmed the ineffectiveness of the use of immunoglobulin for the prevention and treatment of influenza [24], as well as intramuscular administration of drugs for replacement therapy and immunomodulation [21].
In the United States, only one HCI drug is registered for intramuscular administration. Indications for its use are the prevention of hepatitis A, measles, rubella, and chickenpox (in the absence of the possibility of using specific immunoglobulin) [25]. For intramuscular HCI preparations, according to the guidelines of the European Medicines Agency (EMA) [20, 26], there is no requirement to submit data from clinical studies only for the indication “for the prevention of hepatitis A”. For other indications, data from own clinical trials must be submitted. Intramuscular administration of immunoglobulin, according to these documents, is not considered effective for long-term use and is allowed only in exceptional cases.
Intravenous administration of HCI was first used in clinical practice in 1962 [18]. The use of early intravenous HCI preparations was very often accompanied by serious adverse reactions due to the low degree of purification and the content of immunoglobulin A (IgA) in quantities that cause anaphylactic reactions. After the development of a method for processing gamma globulin, preventing the formation of globulin aggregates, purification from proteases, plasmin, plasminogen, prekallikrein activator, and other impurities, it became possible to obtain safe and effective preparations of HCG for intravenous administration. In 1981, the first such drug was registered in the USA. The purpose for its use was to replace the insufficiently effective and painful to use IHCI for intramuscular administration to patients as replacement therapy. Intravenous administration of HCI made it possible to provide the necessary concentration of immunoglobulins, preventing the occurrence of recurrent diseases in the blood of patients with weakened immune systems [27]. After some time, it was found that intravenous administration of IGCH not only successfully replaces intramuscular administration, but also far exceeds it in effectiveness, incl. due to the ability to maintain a certain concentration of immunoglobulins. A clear example of the advantage of intravenous administration of HCI compared with intramuscular administration was the highly effective therapy of idiopathic thrombocytopenic purpura in children [28].
The International Union of Immunological Societies and WHO experts, having summarized the accumulated information on the experience of using normal immunoglobulin in 1982, adopted a memorandum “On the appropriate use of human immunoglobulin in clinical practice,” which listed and justified the criteria for the therapeutic use of immunoglobulin [24]. This document recommends the use of intravenous HCI for the treatment of immunocompromised patients. In addition, emphasis was once again placed on the need to adhere to scientifically based indications for use. It was especially noted that active immunization (vaccination) is more effective compared to passive immunization, which provides protection only for a short time. It is emphasized that it is inappropriate to use HCI for the prevention of infections in premature infants or in the period of physiological hypogammaglobulinemia, or for the treatment of malnutrition (malnutrition).
The text of the document indicates a contraindication to the use of IGNI - IgA deficiency, because. Immunoglobulin therapy can cause the induction of antibodies against IgA and, as a result, an undesirable reaction. However, the use of immunoglobulin may be advisable under specialist supervision in cases of combined IgA-IgG2 deficiency if the drug contains low levels of IgA.
In recent years, HCI drugs have been produced for intravenous administration with high purity, full Fc-fragment activity, and a high degree of viral safety, achieved through a multi-stage production process. At the same time, it should be noted that technological methods have not yet been developed to guarantee 100% viral safety of blood. Only the maximum degree of viral safety has been achieved [22, 29].
The therapeutic effectiveness of immunoglobulins for intravenous administration for a particular pathology is periodically re-evaluated as experience in their use accumulates. The use of standard immunoglobulins for intravenous administration, according to a multicenter study, did not reduce mortality in cardiac surgery patients with severe postoperative systemic inflammatory reaction, which was considered an indication for their use. Long-term clinical practice has shown that only the administration of drugs enriched with immunoglobulins of classes M and A increases the survival of patients in the treatment of sepsis [22].
Currently, 14 HCI drugs for intravenous administration are registered in the Russian Federation (9 of them are foreign-made) [23]. GCI drugs are included in the List of vital and essential drugs for medical use [30]. The analysis showed that only part of the indications in the instructions for use of intravenous HCI drugs produced in Russia coincide with the prescriptions of drugs registered by the EMA and the Food and Drug Administration (FDA) [31, 32]. The instructions for the use of some domestic IHCI drugs for intravenous administration indicate indications that are currently considered insufficiently substantiated and not confirmed by the results of clinical studies: blood diseases, systemic connective tissue diseases, prevention of opportunistic infections and postoperative complications, consequences of immunosuppressive therapy, severe bacterial, including sepsis and viral infections, postoperative complications accompanied by bacteremia and septicopyemic conditions, myasthenia gravis in the acute stage.
Modern technologies for producing immunoglobulin preparations with a high degree of purification have made it possible over the last decade to return to the practice of their subcutaneous administration. The use of immunoglobulin by subcutaneous infusion was discussed in WHO guidelines in 2007 [33]. WHO experts recommended this method of administering IGNI for the treatment of primary immunodeficiencies. Based on many years of practice in the use of subcutaneous administration of HCI, medical practitioners and international control bodies of the European Union indicate in regulatory documents [20, 34, 35] that subcutaneous administration is preferable, because can be carried out at home, allows you to maintain a constant level of IgG in the serum (as opposed to a decrease in the concentration of IgG during intravenous administration), reduces the likelihood of adverse reactions and the transfer of transmissible infectious agents. This method of administration is much more comfortable for patients, often suitable even for patients who have previously experienced adverse reactions with intravenous administration of immunoglobulins, and allows doctors and patients to carry out replacement therapy without premedication with corticosteroids or antihistamines. In addition, subcutaneous use of immunoglobulin is comfortable for both children and adults, as well as for elderly patients and, importantly, significantly reduces the cost of treatment. The infusion duration for subcutaneous immunoglobulin preparations is limited to one hour or less, while intravenous infusions continue for several hours. Currently, subcutaneous IGNIs are mainly used to treat patients with low IgA levels, positive serological inflammatory markers, phlebitis, kidney diseases and other pathologies that create conditions for complications with intravenous administration of immunoglobulins [36]. The goal of replacement therapy is to reduce the frequency and severity of bacterial infections, prevent the development of irreversible and severe complications, as well as life-threatening infections, which is possible when pre-transfusion IgG levels are achieved close to normal [37].
In Europe, over the past 15 years, the demand for immunoglobulin preparations for intravenous and subcutaneous administration has increased 2.5 times [38]. Preparations for subcutaneous administration, taking into account the combination of properties and ease of use, may in the future displace immunoglobulins for intravenous administration from the market. In the Russian Federation, HCI preparations for subcutaneous administration have not yet been registered [23].
In 1991, the first domestic complex immunoglobulin preparation for enteral use was approved for use in healthcare practice. The drug contains immunoglobulins of classes G, M, A, isolated from sediment “B” (fraction III according to Cohn). The content of secretory IgA in the drug reaches 15–25% (for comparison: no more than 3% in other IgChN drugs), which makes it possible to use it topically for infectious and inflammatory processes of the mucous membranes of the gastrointestinal tract. The drug is well tolerated and has the ability to obtain a rapid therapeutic effect, especially in the treatment of dysbacteriosis in children [39]. Currently, 3 HCI drugs for enteral use are registered in the Russian Federation [23]. The drugs are indicated for the treatment of acute intestinal infections in children over 1 month of age.
Demand for human immunoglobulin products is growing as new indications emerge. Thus, at the European Congress of Immunologists (Germany, 2014), it was noted that 42% of global consumption of HCI is associated with neurological indications, 18% with hemato-oncological diseases [38, 40]. In 33% of cases, HCI drugs are used off-label. Currently, numerous clinical studies are being conducted on the use of IGCHN for the treatment of dermatomyositis/polymyositis, chronic idiopathic and complex regional pain syndrome, severe diabetic polyneuropathy, glioblastoma, neuroblastoma, autoimmune autonomic gangliopathy, autoimmune neuropsychiatric disorders in children associated with streptococcal infection, HIV-associated myelopathy, anemia caused by parvovirus B19 and/or cardiomyopathy, spinocerebellar ataxia type 3, acute ischemic stroke, myasthenia gravis, Lambert-Eaton syndrome, toxic epidermal necrolysis, systemic lupus erythematosus and lupus nephritis, idiopathic acute and refractory solar urticaria, in transplantation organs, to prevent antibody-mediated rejection of a transplanted organ, septic shock, pregnant women with primary cytomegalovirus infection, recurrent miscarriages, to remove human leukocyte antigens [38].
In accordance with the approaches of the EMA guidelines, HCI preparations for intravenous administration are recommended for use for the following indications: replacement therapy in adults, children and adolescents (0–18 years) with primary immunodeficiencies with impaired antibody production; hypogammaglobulinemia and recurrent bacterial infections in patients with chronic lymphocytic leukemia when preventive antibiotic therapy is ineffective; hypogammaglobulinemia and recurrent bacterial infections in patients with multiple myeloma in the plateau phase when vaccination with pneumococcal vaccine is ineffective; hypogammaglobulinemia in patients after allogeneic hematopoietic stem cell transplantation; congenital acquired human immunodeficiency syndrome (AIDS) in the presence of recurrent bacterial infections; immunomodulatory therapy for adults, children and adolescents (0–18 years) for idiopathic thrombocytopenic purpura in children or in adults with a high risk of bleeding or before surgery to correct the platelet count, in Guillain–Barre syndrome, in Kawasaki disease [31]. Some drugs, based on the results of their own clinical studies, are approved for use in the treatment of other autoimmune diseases (multifocal motor neuropathy, chronic inflammatory demyelinating polyradiculoneuropathy, myasthenia gravis, etc.). Instructions for use of intravenous HCI drugs registered by the FDA provide similar indications for use [32].
According to the current regulatory documents of the Ministry of Health of Russia and Rospotrebnadzor (orders approving standards for the provision of medical care at various levels, sanitary and epidemiological rules and guidelines for the prevention of certain infectious diseases), IGCHN preparations without specifying the method of administration are recommended for use in children and adults for the prevention of viral hepatitis B, delta, neither A nor B with the parenteral mechanism of pathogen transmission, prevention and treatment of influenza and acute respiratory diseases, prevention and treatment of mumps; children - for the treatment of chickenpox, infectious mononucleosis, cytomegalovirus infection, hemorrhagic fever with renal syndrome, generalized form of meningococcal infection, shigellosis, salmonellosis, acute intestinal infections and food poisoning, pseudotuberculosis, yersiniosis, campylobacteriosis, chronic pancreatitis, gastroenteritis of viral etiology, chronic active hepatitis (autoimmune), liver cirrhosis, Crohn's disease, ulcerative (chronic) ileocolitis (ulcerative colitis), epilepsy, myasthenia gravis complicated by crisis, viral encephalitis, myelitis, acute disseminated encephalomyelitis, juvenile arthritis with systemic onset; adults – for the treatment of polyneuropathy with systemic connective tissue lesions, nodular polyarthritis and related conditions, other necrotizing vasculopathies and other systemic connective tissue lesions, with systemic sclerosis, abscess, boil, skin carbuncle, with induced abortion, with hypertension with significant proteinuria caused pregnancy, patients with unspecified effects of radiation, acute promyelocytic leukemia, hereditary deficiency of factor VIII, hereditary deficiency of factor IX, certain disorders involving the immune mechanism.
Thus, the indications for use and methods of administration of HCIs have undergone significant changes over the 70-year period of their use. The instructions for the use of IGCHN drugs registered in the Russian Federation and other regulatory documents on the treatment and prevention of diseases indicate various indications, which in most cases do not correspond to those for use in other countries; the appropriateness of some of them requires clinical justification. Therefore, it is necessary to update the information in the instructions for the use of immunoglobulin preparations produced in the Russian Federation, unify the indications for the use of drugs with identical routes of administration, and harmonize the information with current information on the experience of using immunoglobulin preparations abroad.
Instructions for Anti-Rhesus Immunoglobulin (Method and dosage)
Before administering the ampoule with the solution, it must be kept at a temperature of 18 to 22 °C for two hours. It cannot be administered intravenously. To prevent the formation of foam, you need to draw the solution into the syringe with a wide-bore needle. An opened bottle cannot be stored.
One dose of the drug is administered intramuscularly once. After childbirth, a woman is given Immunoglobulin during the first three days.
When terminating a pregnancy, the injection should be given immediately after the operation.
The need to administer a certain dose of the drug during a full pregnancy is determined depending on how much fetal blood enters the mother’s bloodstream.
For the purpose of prevention, one dose (300 mcg) of the drug is administered before childbirth; it should be administered at approximately 28 weeks of pregnancy. 2-3 days after birth, another dose of Immunoglobulin is administered, provided that the baby is Rh-positive.
If there is a threat of miscarriage during any period of gestation, one dose of the medicine should be administered.
If a spontaneous abortion or termination of an ectopic pregnancy after the 13th week of gestation, it is recommended to administer 1 dose of the drug. If pregnancy is terminated before the 13th week, a mini-dose (50 mcg) can be administered.
In other cases, the indications and dosage of the drug are determined exclusively by a specialist. It will also help determine which anti-Rhesus Immunoglobulin is better.
Hyper rou s/d (anti-rhesus immunoglobulin) Solution, 9 pcs, 1500 IU
Directions for use and doses
The drug is administered intramuscularly in 1 dose (1500 IU) or 2 doses (3000 IU) once: to a postpartum woman - within 72 hours after birth, in case of termination of pregnancy - immediately after the end of the operation. The following criteria must be met: the mother must be Rh negative and must not already be sensitized to the Rh0(D) factor, her child must be Rh positive. If the drug is given before birth, it is important that the mother receives 1 more dose of the drug after the birth of her Rh-positive baby within 72 hours of delivery. If it is determined that the father is Rh negative, there is no need to administer the drug. Before administration, syringes with the drug are kept for 2 hours at room temperature (20±2°C). In order to avoid the formation of foam, the drug is drawn into a syringe with a wide-bore needle. For prophylaxis in the postpartum period, 1 dose (1500 IU) of HyperROU S/D should be administered within the first 72 hours after birth. The need for a certain dose in the case of a full term pregnancy varies depending on the volume of fetal blood entering the mother's bloodstream. 1 dose (1500 IU) contains a sufficient amount of antibodies to prevent sensitization to the Rh factor, if the volume of fetal red blood cells entering the bloodstream does not exceed 15 ml. In cases where a larger volume of fetal red blood cells (more than 30 ml of whole blood or more than 15 ml of red blood cells) is expected to enter the maternal circulation, a fetal red blood cell count should be performed using an approved laboratory technique (for example, a modified acid washout-stain method according to Kleihauer and Betke ) to determine the required dose of the drug. The calculated volume of fetal red blood cells entering the mother's bloodstream is divided by 15 ml and the number of doses of HyperROU S/D that must be administered is obtained. If the presence of more than 15 ml of fetal red blood cells is expected or the dose calculation results in a fraction, the number of doses should be rounded up to the next whole number upward, for example, if a result of 1.4 is obtained, 2 doses (3000 IU) of the drug should be administered. For prophylaxis during the prenatal period, 1 dose of the drug (1500 IU) should be administered at approximately the 28th week of pregnancy. This must be followed by 1 more dose (1500 IU), preferably within 72 hours after birth, if the newborn baby is Rh positive. If pregnancy continues after the threat of abortion arises, at any stage of pregnancy, 1 dose (1500 IU) of the drug should be administered. If it is suspected that more than 15 ml of fetal red blood cells have entered the maternal bloodstream, the dose must be adjusted (as indicated above). After spontaneous abortion, induced abortion or termination of an ectopic pregnancy during pregnancy more than 13 weeks, it is recommended to administer 1 dose (1500 IU) of the drug. If it is suspected that more than 15 ml of fetal red blood cells have entered the maternal bloodstream, the dose must be adjusted (as indicated above). If the pregnancy is terminated at less than 13 weeks, a single mini-dose of HyperROU S/D (approximately 250 IU) may be administered. After amniocentesis at the 15-18th week of pregnancy, or during the third trimester of pregnancy, or if an injury to the abdominal organs occurs during the second and/or third trimester of pregnancy, 1 dose (1500 IU) of the drug should be administered. If more than 15 ml of red blood cells are suspected of entering the mother's bloodstream, the dose must be adjusted as described above. If abdominal trauma, amniocentesis, or other unfavorable circumstance requires administration of the drug at 13-18 weeks of pregnancy, another 1 dose (1500 IU) should be administered at 26-28 weeks. To maintain protection throughout pregnancy, the concentration of passively obtained antibodies to Rh0(D) should not fall below the level necessary to prevent an immune response to Rh-positive red blood cells. T1/2 of human anti-rhesus immunoglobulin Rh0(D) is 23-26 days. In any case, the dose of the drug should be administered within 72 hours after birth if the baby is Rh positive. If delivery occurs within 3 weeks of the last dose, the postpartum dose can be discontinued (unless more than 15 ml of fetal red blood cells have entered the maternal circulation).
Anti-Rhesus Immunoglobulin during pregnancy and lactation
If indicated, the drug is administered to women during pregnancy , and also, if necessary, after childbirth . In this case, the instructions for pregnancy, which contain information about the dosage of the drug, must be strictly followed. Reviews from women indicate that the drug, as a rule, does not cause significant side effects. Whether it is possible to receive anti-Rhesus Immunoglobulin during pregnancy for free, you need to find out at a specific medical institution.
Human immunoglobulin anti-rhesus Rho(D) solution IM 300 mcg/dose 1 ml x1
ATX code: J06BB01 (Anti-D(rh) immunoglobulin) Active substance: human anti-D immunoglobulin Ph.Eur. European Pharmacopoeia Instructions for use:
HUMAN IMMUNOGLOBULIN ANTI-RHESUS Rho (D)
Registration certificate 003599/01 dated 28.02010.
Trade name of the drug. Human anti-rhesus immunoglobulin Rho (D)
INN or group name. Human immunoglobulin anti-rhesus Rho (D)
Dosage form: solution for intramuscular administration.
Compound.
Active substance: protein immunoglobulin -10%
Excipient: stabilizer - glycine (aminoacetic acid) 2%, water for injection.
Description. Transparent or slightly opalescent liquid, colorless or light yellow. A slight sediment may appear, which disappears when shaken.
Pharmacotherapeutic group: Immunoglobulins
ATX code: [J06BB01]
Pharmacological properties.
The drug is an immunologically active protein fraction isolated from human plasma or serum of donors tested for the absence of antibodies to the human immunodeficiency virus (HIV-1, HIV-2), hepatitis C virus and hepatitis B virus surface antigen. The active component of the drug is immunoglobulin G , containing incomplete anti-Rho(D) antibodies. The drug prevents Rh sensitization (formation of Rho (D)
antibodies) during pregnancy in Rh-negative women who gave birth to Rho (D) positive children or who underwent artificial termination of pregnancy with Rho (D) positive husband’s blood.
Pharmacokinetics.
The maximum concentration of antibodies in the blood after intramuscular injection of human anti-rhesus immunoglobulin Rho(D) is achieved after 24 hours; the half-life of antibodies from the body is 4-5 weeks.
Indications for use.
The drug is used only as prescribed by a doctor.
Human anti-Rhesus immunoglobulin Rho(D) is used to prevent Rh-conflict in Rh-negative women who are not sensitized to the Rho(D) antigen (i.e. have not developed Rh antibodies) under the condition: pregnancy and birth of a Rh-positive child, with artificial and spontaneous abortion, in case of termination of an ectopic pregnancy, in case of a threat of termination of pregnancy at any stage, after amniocentesis and other procedures associated with the risk of fetal blood entering the mother’s bloodstream, as well as in the event of an abdominal injury.
Contraindications.
Administration of human anti-Rhesus immunoglobulin Rho(D) is contraindicated in Rh-positive postpartum women, Rh-negative postpartum women, sensitized to the Rho (D) antigen (in whose blood serum Rh antibodies are detected).
Method of administration and dose.
Human anti-rhesus immunoglobulin Rho(D) is used intramuscularly only. Before injection, ampoules with the drug are kept for 2 hours at room temperature (20±2) °C. To avoid the formation of lena, the drug is drawn into a syringe with a wide-bore needle. The drug cannot be stored in an opened ampoule.
One dose (300 mcg) corresponds to 1 ml with an antibody titer of 1: 2000 or 2 ml with an antibody titer of 1: 1000.
During pregnancy, Rh-negative women without sensitization phenomena undergo nonspecific desensitizing therapy at 10-12, 24-25, 32-33 weeks of pregnancy. Human anti-rhesus immunoglobulin Rho(D) is administered in one dose (300 mcg) intramuscularly. In case of Rh(+) (positive Rh factor) in the husband's blood, administration of anti-Rhesus immunoglobulin Rho(D) 300 mcg intramuscularly at 28-30 weeks. Repeated administration of immunoglobulin no later than 48-72 hours after birth at the birth of an Rh-positive child.
In case of artificial termination of pregnancy during pregnancy more than 13 weeks, it is recommended to administer one dose of the drug (300 mcg).
If pregnancy is terminated at less than 13 weeks, it is recommended to administer 50 mcg of the drug.
In case of injury to the abdominal organs during the second or third trimester of pregnancy, it is recommended to administer one dose of the drug (300 mcg). If abdominal trauma requires administration of the drug at 13-18 weeks' gestation, another dose (300 mcg) should be administered at 26-28 weeks' gestation.
Side effect.
In rare cases, reactions may develop in the form of skin hyperemia at the injection site and an increase in temperature to 37.5 ° C during the first day after administration, as well as dyspepsia. Individuals with altered reactivity may develop allergic reactions of various types, and in exceptional cases, anaphylactic shock, therefore persons receiving the drug should be monitored for 30 minutes. Antishock therapy must be available in the room where the drug is administered.
Interaction with other drugs.
Immunoglobulin therapy can be combined with other drugs, in particular antibiotics.
Special instructions.
Immunization of women with live vaccines should be carried out no earlier than 3 months after the administration of human anti-rhesus immunoglobulin Rho(D). Cannot be administered intravenously.
Release form.
Solution for intramuscular administration 300 mcg/dose. 1.0 ml (300 mcg) of the drug with an antibody titer of at least 1: 2000 or 2.0 ml (300 mcg) with an antibody titer of 1: 1000 in ampoules. 1 or 10 ampoules along with instructions for use and an ampoule knife are packaged in a box of cardboard.
Storage conditions.
The drug is stored in a dry place, protected from light at a temperature of 2 to 10 ° C. Preparations in ampoules with damaged integrity or labeling, changes in physical properties (change in color, clouding of the solution, presence of non-developing flakes), expired, or improper storage are not suitable for use. Keep out of the reach of children.
Transportation is carried out by any type of covered transport at a temperature of 2 to 10 °C.
Best before date. 3 years.
The drug should not be used after the expiration date.
Conditions for dispensing from pharmacies. On prescription.
Manufacturer. OBUZ "Ivanovo Regional Blood Transfusion Station".