Pharmacological properties of the drug Regulax picosulfate drops
Sodium picosulfate is a contact laxative that irritates receptors in the intestinal mucosa. Activated in the large intestine under the influence of bacterial sulfatases; the substance that is released stimulates the sensitive nerve endings of the intestinal mucosa, enhancing its motility. The laxative effect of sodium picosulfate is not accompanied by tenesmus and intestinal spasms. In infants, the effectiveness of sodium picosulfate may be insufficient due to the small amount of bacterial flora that produces sulfatases. Regulax Picosulfate drops are a drug that regulates intestinal function, increasing the volume of stool and enhancing peristalsis (natural rhythmic movement), promoting timely, painless bowel movements. The laxative effect occurs after 10–12 hours. Sodium picosulfate is absorbed in the small intestine only in limited quantities and has virtually no systemic effect. After the sulfate ester is broken down by bacteria in the large intestine, a diphenolic laxative is formed, which is partially absorbed and, after conjugation, is excreted in the bile. A small portion is excreted in the urine as a glucuronide; most of it is in the feces, both in the form of free diphenol and in the form of picosulfate itself.
Regulax Picosulfate drops for internal use 20ml
Compound
Active substance: sodium picosulfate monohydrate 7.5 mg.
Excipients: sorbitol solution 70% (non-crystallizing) - 651.4 mg, propylene glycol - 200 mg, purified water - 308 mg.
Pharmacokinetics
After oral administration, it is not absorbed from the gastrointestinal tract and does not undergo hepatic-intestinal circulation.
Indications for use
- Atonic constipation;
- regulation of stool (hemorrhoids, proctitis, anal fissures);
- preparation for surgical operations, instrumental and x-ray examinations.
Contraindications
- Hypersensitivity to the drug;
- intestinal obstruction;
- strangulated hernia;
- acute inflammatory diseases of the abdominal organs;
- peritonitis;
- abdominal pain (of unknown origin);
- bleeding from the gastrointestinal tract;
- metrorrhagia;
- cystitis;
- severe dehydration;
- spastic constipation;
- children under 4 years of age;
- I trimester of pregnancy.
With caution: lactation period (breastfeeding).
Directions for use and doses
Inside, before bedtime. Depending on the effect obtained, the dose at subsequent doses is increased or decreased. Adults: initial dose - 13 drops, for persistent constipation - up to 26 drops. For children over 4 years old, the initial dose is 5-8 drops. The course of treatment is 7 days.
Storage conditions
The drug should be stored out of the reach of children at a temperature of 15° to 25°C.
Best before date
5 years.
Do not use after the expiration date stated on the package. Shelf life after opening the bottle is 6 months.
special instructions
Should not be taken without medical supervision for more than 7 days. Long-term use often leads to increased intestinal hardening.
Should be used in children only with the advice of a doctor.
Instructions for patients with diabetes mellitus
Suitable for diabetics; contains sugar substitutes; 1 ml corresponds to 0.03 XE.
Description
Laxative.
Use in children
Should be used in children only with the advice of a doctor.
Contraindicated in children under 4 years of age.
Pharmacodynamics
Laxative. The active form of the drug, formed by hydrolysis under the influence of intestinal microorganisms, directly stimulates the nervous structures of the intestinal wall, as a result of which the movement of intestinal contents accelerates and the absorption of electrolytes and water decreases. The effect occurs 10-12 hours after administration.
Side effects
Possible: diarrhea, abdominal pain, bloating, dehydration, water and electrolyte imbalance, weakness, cramps, decreased blood pressure.
Use during pregnancy and breastfeeding
Use is contraindicated in the first trimester of pregnancy. Repeated use of the drug during the second and third trimesters should be carried out only after a careful assessment of the need and risk, since there is not enough information on the use of the drug during pregnancy.
Interaction
Possible increased sensitivity to cardiac glycosides.
GCS and diuretics increase the risk of developing electrolyte disturbances.
Broad-spectrum antibiotics reduce the laxative effect.
Overdose
Symptoms: with chronic overdose, ischemia of the colon mucosa, secondary hyperaldosteronism, urolithiasis, and damage to the renal tubules may develop.
Treatment: gastric lavage, correction of water-electrolyte imbalance, antispasmodics.
Indications for use of the drug Regulax picosulfate drops
Intestinal atony, constipation (except for spastic constipation), caused by changes in the nature and diet, prolonged immobilization, fever, severe general condition, diseases of the cardiovascular system and metabolic disorders; used in the pre- and postoperative periods, in obstetric and gynecological practice, to facilitate defecation in case of hemorrhoids, anal fissures, inoperable hernias, myocardial infarction, severe hypertension (arterial hypertension), to cleanse the intestines before instrumental examination.
Regulax picosulfate drops for oral administration 7.5 mg/ml 20 ml No. 1
Name
Regulax picosulfate for oral administration 7.5 mg/ml in vial. 20ml per pack. No. 1
Description
Drops for oral administration
Main active ingredient
Sodium picosulfate
Release form
Drops
Dosage
7.5 mg/ml
special instructions
Check with your doctor before you start taking Regulax® Picosulfate, oral drops. — In children over 4 years of age, Regulax® Picosulfate. Oral drops should be used only as prescribed by a doctor. Be especially careful if constipation occurs suddenly, if you have been constipated for a long time and/or if there is blood in your stool, or if constipation is accompanied by a fever. In such cases, you should consult your doctor and undergo an examination before starting to use Regulax® Picosulfate for oral administration, since stool disorders may be a sign of a serious illness. Regulax® Picosulfate, oral drops should not be taken for constipation every day or for a long period without consulting a doctor. Symptoms may reappear after stopping Regulax® Picosulfate, oral drops. After taking for chronic constipation for a long period, return of symptoms may be due to worsening constipation. Patients taking medications containing sodium picosulfate have reported dizziness and fainting. However, available information suggests that syncope was caused by straining to defecate or by cardiovascular reactions to abdominal pain. If you have an intolerance to certain sugars, please consult your doctor before taking Regulax® Picosulfate, oral drops.
pharmachologic effect
Laxative. The active form of the drug, formed by hydrolysis under the influence of intestinal microorganisms, directly stimulates the nervous structures of the intestinal wall, as a result of which the movement of intestinal contents accelerates and the absorption of electrolytes and water decreases. The effect occurs 10-12 hours after administration.
Pharmacokinetics
After oral administration, it is not absorbed from the gastrointestinal tract and does not undergo hepatic-intestinal circulation.
Indications for use
- atonic constipation; - regulation of stool (hemorrhoids, proctitis, anal fissures); — preparation for surgical operations, instrumental and x-ray examinations.
Directions for use and doses
Regulax® Picosulfate, oral drops should always be used in accordance with these instructions for use. If you have any doubts, contact your doctor or pharmacist. Recommended dosage (unless otherwise prescribed by your doctor): Adults and adolescents should take 14-27 drops of Regulax® Picosulfate (corresponding to 5-10 mg sodium picosulfate). Children 4 years of age and older should take Regulax® Picosulfate only as prescribed by a doctor, 7-14 drops of the drug (corresponding to 2.5-5 mg of sodium picosulfate). Directions for use: Regulax® Picosulfate, oral drops, preferably taken in the evening. Drops can be taken alone or with water. The laxative effect of the drug usually occurs within 10-12 hours. Please take into account the time of onset of action of the drug; you should not push to induce bowel movements. Duration of use: Regulax® Picosulfate, oral drops should not be used daily for an extended period or for an extended period without consulting a doctor. If you feel that the effect of Regulax® Picosulfate Oral Drops is too strong or too weak, discuss this with your doctor or pharmacist. If you take more Regulax® Picosulfate Oral Drops than you should: Taking high doses may cause diarrhea, complaints of abdominal pain, dehydration, loss of potassium and other minerals. Treatment of overdose is symptomatic. In case of acute overdose within a short time after administration, absorption of the drug can be reduced or prevented by inducing vomiting or gastric lavage. Measures should be taken in accordance with the doctor’s instructions to restore large losses of fluid and electrolytes. In some cases, it may make sense to use antispasmodics. In addition, there are reports of isolated cases of reduction or cessation of blood supply to the colon mucosa, in which doses of sodium picosulfate were significantly higher than the doses recommended for the treatment of constipation. Note: It is well known that chronic overdose of laxatives, including Regulax® Picosulfate, oral drops, can lead to chronic diarrhea, abdominal pain, hypokalemia, secondary hyperaldosteronism and kidney stones. Cases of renal tubular damage, metabolic alkalosis, and muscle weakness due to hypokalemia have also been reported in association with chronic laxative abuse.
Use during pregnancy and lactation
If you are pregnant, breastfeeding, think you are pregnant, or plan to become pregnant, tell your doctor or pharmacist before taking this medicine. Pregnancy There are no clinical data on the use of sodium picosulfate during pregnancy. It is recommended to avoid the use of sodium picosulfate during pregnancy. Breastfeeding Studies have shown that the active substance does not pass into breast milk. Therefore, Regulax® Picosulfate, drops for oral administration can be used during breastfeeding.
Precautionary measures
If you have an intolerance to certain sugars, please consult your doctor before taking Regulax® Picosulfate, oral drops.
Interaction with other drugs
Tell your doctor or pharmacist if you are taking, have recently taken or plan to take other medicines. — Antibiotics (medicines that inhibit the growth of bacteria) can weaken or completely neutralize the effect of Regulax® Picosulfate, drops for oral administration. — When taking large doses of Regulax® Picosulfate, oral drops, there is a risk of an imbalance of potassium and other electrolytes in the blood. If you are taking medications that also remove potassium from the body, such as some diuretics (diuretics) and corticosteroids (adrenal hormones), this may cause hypokalemia with impaired heart function and muscle weakness and increased sensitivity to cardiac glycosides, which stimulate the heart. activity.
Contraindications
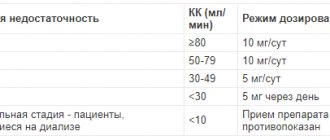
- If you are allergic to sodium picosulfate, triarylmethane derivatives or other components of the drug listed in the “Composition” section. — With narrowing of the intestinal lumen with impaired passage of intestinal masses, intestinal obstruction, acute abdominal conditions (for example, appendicitis), with intense abdominal pain accompanied by nausea and vomiting, — With acute inflammatory diseases of the gastrointestinal tract, — With severe dehydration of the body. - For bleeding from the rectum of unknown origin, stenosis of the intestine or biliary tract, cholelithiasis, liver failure. Children under 4 years of age should not take Regulax® Picosulfate, drops for oral administration. Constipation, accompanied by ailments such as abdominal pain, vomiting and fever, may be a sign of a serious illness (intestinal obstruction, acute inflammation of the abdominal organs). If you have such complaints, you should not take Regulax® Picosulfate, oral drops or other medications, but should consult a doctor immediately. For diseases accompanied by disturbances in water and electrolyte balance (for example, limited renal function), Regulax® Picosulfate, oral drops can only be used under medical supervision.
Compound
Active ingredient: sodium picosulfate. 1 ml of solution (20 drops) contains 7.5 mg of sodium picosulfate monohydrate (corresponding to 7.23 mg of sodium picosulfate). Other ingredients: sorbitol solution 70% (non-crystallizing), propylene glycol, purified water. Instructions for patients with diabetes mellitus: The drug is suitable for patients with diabetes mellitus. The drug contains a sugar substitute (1 ml corresponds to 0.03 XE (bread units)).
Overdose
Symptoms: with chronic overdose, ischemia of the colon mucosa, secondary hyperaldosteronism, urolithiasis, and damage to the renal tubules may develop. Treatment: gastric lavage, correction of water-electrolyte imbalance, antispasmodics.
Side effect
Like other medicines, Regulax® Picosulfate Oral Drops can cause side effects, although not everyone gets them. The incidence of the adverse reactions listed below is estimated as follows: Very common: more than 1 in 10 patients Common: 1 to 10 in 100 patients Uncommon: 1 to 10 in 1,000 patients Rare: 1 to 100 patients 10 in 10,000 patients Very rare: less than 1 in 10,000 patients Not known: It is not possible to determine the incidence of the adverse reaction based on available data. Possible adverse reactions: From the immune system Not known: allergic reactions (including skin reactions: swelling of the skin and/or mucous membrane (angioedema), for example, in the face and throat, possibly difficulty breathing). From the nervous system: Uncommon: dizziness. Unknown: fainting. Available information suggests that these ailments are a consequence of straining during bowel movements or a reaction of the cardiovascular system to abdominal pain (see section “The drug should not be used”). From the gastrointestinal tract Very often: diarrhea. Common: flatulence, abdominal pain or abdominal colic. Uncommon: nausea, vomiting. Taking Regulax® Picosulfate, drops for oral administration over a long period or using high doses is often accompanied by an increase in the excretion of water, potassium and other salts from the body. This may lead to cardiac dysfunction and muscle weakness, especially when taken concomitantly with diuretics or corticosteroids. In case of severe manifestation of one of the listed side reactions or side effects not listed in these instructions, inform your doctor.
Storage conditions
Store at a temperature of 15°C to 25°C out of the reach of children.
Special instructions for the use of Regulax picosulfate drops
The drug Regulax Picosulfate drops is intended for short-term use. During pregnancy, the use of the drug is possible only under strict indications and under the supervision of a doctor. While using the drug, you should stop breastfeeding. The drug can be used for diabetes mellitus, since it contains a sugar substitute (1 ml of drops contains 0.03 XE). The drug should be used with caution after operations on the gastrointestinal tract or in case of dehydration. No addiction was observed with long-term use.
Regulax Picosulfate (vial 7.5 mg/ml 20 ml)
A country
Germany
The country of production may vary depending on the batch of goods. Please check with the operator for detailed information when confirming your order.
Active substance
Sodium picosulfate
Compound
Active substance: sodium picosulfate monohydrate 7.5 mg.
pharmachologic effect
Laxative. Reduces the absorption of electrolytes and water, enhances intestinal motility. The active form of the drug, formed by hydrolysis under the influence of intestinal microorganisms, directly excites the nervous structures of the intestinal wall, resulting in accelerated movement of intestinal contents. The action occurs 10-12 hours after administration. Pharmacokinetics. After oral administration, it is not absorbed from the gastrointestinal tract and is not subjected to hepatic-intestinal circulation.
Indications for use
Atonic constipation. Regulation of stool (hemorrhoids, proctitis, anal fissures). Preparation for surgical operations, instrumental and x-ray examinations.
Mode of application
Inside, before bedtime. Depending on the effect obtained, the dose at subsequent doses is increased or decreased. Adults: initial dose - 13 drops, for persistent constipation - up to 26 drops. For children over 4 years old, the initial dose is 5-8 drops. The course of treatment is 7 days.
Interaction
Increased sensitivity to cardiac glycosides is possible. Glucocorticosteroids and diuretics increase the risk of developing electrolyte disturbances. Broad-spectrum antibiotics reduce the laxative effect. Possible increased sensitivity to cardiac glycosides.
Side effect
Diarrhea, abdominal pain, dehydration, water and electrolyte imbalance, weakness, cramps, decreased blood pressure.
Contraindications
Hypersensitivity, intestinal obstruction, strangulated hernia, acute inflammatory diseases of the abdominal organs. Peritonitis, abdominal pain (of unknown origin), severe dehydration, bleeding from the gastrointestinal tract, metrorrhagia, cystitis, spastic constipation, children (up to 4 years), pregnancy (1st trimester).
Overdose
Symptoms: see section "Side effects". In addition, with chronic overdose, ischemia of the colon mucosa, secondary hyperaldosteronism, urolithiasis, and damage to the renal tubules may develop. Treatment: gastric lavage, correction of water-electrolyte imbalance, antispasmodics.
special instructions
With caution: lactation period. Use during pregnancy. Contraindicated in the first trimester of pregnancy. Repeated use of the drug during the second and third trimesters should be carried out only after a thorough assessment of the need and risks, since there is not enough information on the use of the drug during pregnancy. It should not be taken without medical supervision for more than 7 days. Long-term use often leads to increased intestinal hardening. Should be used in children only in consultation with a doctor.Instructions for patients with diabetes.Suitable for patients with diabetes; contains sugar substitutes.