Pharmacological properties of the drug Guttalax
Sodium picosulfate is a local laxative of the triarylmethane group, which, after bacterial breakdown in the large intestine, stimulates its mucous membrane, improving peristalsis. After oral administration, sodium picosulfate reaches the large intestine without significant absorption and therefore does not enter the enterohepatic circulation. As a result of bacterial breakdown of the starting substance in the large intestine, free diphenol is formed. The effect develops after 6–12 hours. Only a small amount of sodium picosulfate can be detected in tissues after oral administration. The use of the drug reflexively reduces the systemic load on the urinary system. The laxative effect of the drug does not depend on the concentration of active diphenol in the blood plasma.
Guttalax drops 7.5 mg/ml fl 15 ml
Compound
Active substance: sodium picosulfate monohydrate - 0.75 g. Excipients: sodium benzoate - 0.2 g, sodium citrate dihydrate - 0.15 g, liquid sorbitol (non-crystallizing) - 64.37 g, citric acid monohydrate - 0.14 g, purified water - 49.89 g.
Pharmacokinetics
Suction and distribution
After oral administration of the drug, sodium picosulfate enters the colon without significant absorption. Thus, enterohepatic circulation of the drug is excluded.
Metabolism
In the distal colon, bacterial breakdown of sodium picosulfate occurs to form the active metabolite bis-(p-hydroxyphenyl)-pyridyl-2-methane (BHPM), which has a laxative effect.
Removal
After cleavage, a small amount of BGPM is absorbed and then almost completely binds in the intestinal wall and liver to form an inactive glucuronide. After oral administration of sodium picosulfate at a dose of 10 mg (about 10.4% of the total dose), the drug is excreted in the urine in the form of BGPM glucuronide after 48 hours. When sodium picosulfate is used in higher doses, its excretion by the kidneys decreases.
Pharmacokinetics/pharmacodynamics relationship
The time for development of the laxative effect of the drug is determined by the rate of release of the active metabolite (BGPM) and is 6-12 hours after use (on average 10 hours).
A small part of the drug enters the systemic circulation. There is no relationship between the laxative effect of the active metabolite and its concentration in the blood serum.
Indications for use
As a laxative in the following cases:
- constipation caused by atony and hypotension of the colon (including in old age, in bedridden patients, after operations, after childbirth and during lactation);
- constipation caused by taking medications;
- to regulate stool in case of hemorrhoids, proctitis, anal fissures (to soften the consistency of stool);
- gallbladder diseases, irritable bowel syndrome with a predominance of constipation;
- constipation caused by intestinal dysbiosis and diet disorders.
Contraindications
- intestinal obstruction;
- obstructive bowel diseases;
- acute diseases of the abdominal organs or severe abdominal pain, which may be accompanied by nausea, vomiting, fever (including appendicitis);
- acute inflammatory bowel diseases;
- severe dehydration;
- fructose intolerance;
- hypersensitivity to sodium picosulfate or other components of the drug.
Directions for use and doses
- The drug is taken orally. The drops do not have to be dissolved in liquid.
- To obtain a laxative effect in the morning, you should take the drug the night before.
- Adults and children over 10 years of age are prescribed 10-20 drops (5-10 mg)/day. The maximum daily dose is 20 drops (10 mg).
- Children aged 4-10 years are prescribed 5-10 drops (2.5-5 mg)/day. The maximum daily dose is 10 drops (5 mg).
- The recommended dose for children under 4 years of age is 0.25 mg/kg body weight/day. This corresponds to 1 drop of the drug (0.5 mg sodium picosulfate) per 2 kg of body weight/day.
- It is recommended to start treatment with a lower dose. In order to achieve regular bowel movements, the dose can be increased to the maximum recommended. The maximum recommended daily dose should not be exceeded.
- Because the drug is not addictive, the dose can be individually adjusted by the patient: the dose can be reduced depending on individual needs or the drug can be used once as needed.
Storage conditions
The drug should be stored out of the reach of children at a temperature not exceeding 30°C, and do not freeze.
Best before date
3 years. Do not use after the expiration date stated on the package.
special instructions
For many patients suffering from constipation, it is not necessary to take the full dose daily; the dose can be reduced depending on the patient's individual needs or the drug can be used as a single dose as needed.
To restore the natural rhythm of bowel movements, along with taking a laxative, it is recommended to increase physical activity, sufficient intake of dietary fiber (20-25 g/day) and fluid (at least 2 l).
You should not use the drug daily without consulting a doctor for more than 10 days. Long-term use of the drug in high doses can lead to fluid loss, electrolyte imbalance, and hypokalemia.
Dizziness and fainting have occurred in patients taking Guttalax®. The analysis showed that these cases are associated with syncope with defecation (or syncope caused by straining to defecate) or with a vasovagal response to abdominal pain, which may be due to constipation, and is not necessarily related to the drug.
1 ml of drops contains 0.45 mg of sorbitol. The maximum recommended daily dose for the treatment of adults and children 4-10 years old contains 0.6 g and 0.3 g of sorbitol, respectively.
Use in pediatrics
Children should take the drug only as prescribed by a doctor.
The drug has no taste, so it can be added to children's food.
Description
Laxative drug.
Dosage form
Drops for oral administration in the form of a clear, colorless to yellowish or slightly yellowish-brown, slightly viscous solution.
Action
The active substance, sodium picosulfate, is a laxative of the triarylmethane group.
As a local laxative, sodium picosulfate, after bacterial breakdown in the large intestine, has a stimulating effect on the mucous membrane of the large intestine, increasing peristalsis, promoting the accumulation of water and electrolytes in the large intestine. This leads to stimulation of the act of defecation, reduction of evacuation time and softening of the stool.
Sodium picosulfate, being a laxative acting at the level of the colon, stimulates the natural process of evacuation of contents from the lower gastrointestinal tract. Therefore, sodium picosulfate does not affect the digestion or absorption of high-calorie foods or essential nutrients in the small intestine.
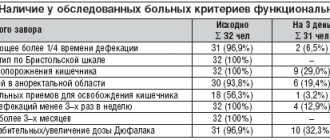
A randomized, double-blind, parallel group study involving 367 patients with chronic constipation examined the effects of Guttalax®. Study results showed a significant increase in intestinal motility within a week compared to placebo for all 4 weeks of therapy (p<0.0001). Serum potassium concentration levels were unchanged at the end of the study (4.4 mM) and corresponded to physiological norms (3.6-5.3 mM).
Side effects
From the digestive system: discomfort, nausea, vomiting, cramps and pain in the abdomen, diarrhea.
From the nervous system: dizziness and fainting. Dizziness and fainting that occur after taking the drug appear to be associated with a vasovagal response (eg, straining during bowel movements, abdominal cramps).
From the immune system: hypersensitivity reactions.
From the skin and subcutaneous tissues: skin reactions, for example, angioedema, skin rash, itching.
Use during pregnancy and breastfeeding
There are no reliable and well-controlled studies in pregnant women. Long-term experience with use has not revealed a negative effect of the drug on pregnancy. Taking the drug in the first trimester of pregnancy is contraindicated. In the second and third trimesters of pregnancy (as with the use of other laxatives), the use of the drug is possible only as prescribed by a doctor.
Studies on the effect of the drug on fertility have not been conducted. In preclinical studies, no teratogenic effects on reproduction were identified.
Interaction
Diuretics or corticosteroids increase the risk of electrolyte imbalance (hypokalemia) when using the drug in high doses.
Electrolyte imbalance may increase sensitivity to cardiac glycosides.
The combined use of the drug and antibiotics may reduce the laxative effect of the drug.
Overdose
Symptoms
When using the drug in high doses, diarrhea, dehydration, decreased blood pressure, water and electrolyte imbalance, hypokalemia, and convulsions are possible. In addition, there are reports of cases of ischemia of the muscles of the large intestine associated with taking the drug in doses significantly higher than those recommended for the usual treatment of constipation.
The drug Guttalax®, like other laxatives, in chronic overdose can lead to chronic diarrhea, abdominal pain, hypokalemia, secondary hyperaldosteronism, and urolithiasis. Renal tubular damage, metabolic alkalosis, and muscle weakness associated with hypokalemia may develop due to chronic laxative abuse.
Treatment
To reduce the absorption of the drug after oral administration, you can induce vomiting or perform gastric lavage. Fluid replacement and correction of electrolyte balance may be required, as well as antispasmodics.
Impact on the ability to drive vehicles and operate machinery
There have been no special clinical studies of the effect of the drug on the ability to drive vehicles or operate machinery. Despite this, patients should be advised that when using the drug, dizziness and/or fainting may occur due to a vasovagal reaction (i.e. during intestinal spasm). If intestinal spasms occur, patients should avoid potentially hazardous activities, incl. driving vehicles or operating machinery.
Special instructions for the use of the drug Guttalax
Long-term studies have not revealed any negative effects of the drug when administered during pregnancy, however, like other drugs, it can be prescribed to pregnant women only as prescribed by a doctor. Despite the lack of data on the penetration of Guttalax into breast milk, the drug should not be prescribed during breastfeeding. Like other laxatives, Guttalax is not recommended for continuous use for a long time. Long-term use of the drug in a high dose can lead to the development of water and electrolyte imbalance (including hypokalemia).
Guttalax laxative for constipation tablets No. 20
A country
France
The country of production may vary depending on the batch of goods. Please check with the operator for detailed information when confirming your order.
Active substance
Sodium picosulfate
Compound
1 tablet contains: sodium picosulfate monohydrate 5.187 mg. Excipients: lactose monohydrate - 71 mg, corn starch - 41.5 mg, colloidal silicon dioxide - 1.7 mg, hydrolyzed potato starch - 0.3 mg, magnesium stearate - 0.5 mg. The tablets are white, round, flat-cylindrical, with beveled edges, on one side of the tablet “5L” is engraved on both sides of the dividing line, on the other side there is the company logo, practically odorless.
pharmachologic effect
The active ingredient - sodium picosulfate - is a laxative of the triarylmethane group. As a local laxative, sodium picosulfate, after bacterial breakdown in the large intestine, has a stimulating effect on the mucous membrane of the large intestine, increasing peristalsis, promoting the accumulation of water and electrolytes in the large intestine. This leads to stimulation of the act of defecation, reduction of evacuation time and softening of the stool. Sodium picosulfate, being a laxative acting at the level of the colon, stimulates the natural process of evacuation of contents from the lower gastrointestinal tract. Therefore, the drug does not affect the digestion or absorption of high-calorie foods or essential nutrients in the small intestine.
Indications for use
As a laxative in the following cases: - constipation caused by atony and hypotension of the colon (including in old age, in bedridden patients, after surgery, after childbirth and during lactation); - constipation caused by taking medications; - to regulate stool in case of hemorrhoids, proctitis, anal fissures (to soften the consistency of stool); - gallbladder diseases, irritable bowel syndrome with a predominance of constipation; - constipation caused by intestinal dysbiosis, dietary disorders.
Mode of application
The drug is taken orally. The tablets must be taken with a sufficient amount of liquid. Drops do not have to be dissolved in liquid. To obtain a laxative effect in the morning, you should take the drug the night before. Adults and children over 10 years old are prescribed 1-2 tablets. or 10-20 drops (5-10 mg)/day. Children aged 4-10 years are prescribed 1/2-1 tablet. or 5-10 drops (2.5-5 mg)/day. Children under 4 years of age should not use the drug in tablet form; it is recommended to prescribe the drug in the form of drops. The recommended dose for children under 4 years of age is 0.25 mg/kg body weight/day. This corresponds to 1 drop of the drug (0.5 mg sodium picosulfate) per 2 kg of body weight/day. It is recommended to start treatment with a lower dose. In order to achieve regular bowel movements, the dose can be increased to the maximum recommended. The maximum recommended daily dose should not be exceeded.
Interaction
Diuretics or corticosteroids increase the risk of electrolyte imbalance (hypokalemia) when using the drug in high doses. Electrolyte imbalance may increase sensitivity to cardiac glycosides. The combined use of the drug and antibiotics may reduce the laxative effect of the drug.
Side effect
From the digestive system: discomfort, nausea, vomiting, cramps and pain in the abdomen, diarrhea. From the nervous system: dizziness and fainting. Dizziness and fainting that occur after taking the drug appear to be associated with a vasovagal response (for example, straining during bowel movements, abdominal cramps). From the immune system: hypersensitivity reactions, including angioedema, skin rash, itching.
Contraindications
- intestinal obstruction; - obstructive bowel diseases; - acute diseases of the abdominal organs or severe abdominal pain, which may be accompanied by nausea, vomiting, fever (including appendicitis); - acute inflammatory bowel diseases; - severe dehydration; - lactase deficiency, lactose intolerance, glucose-galactose malabsorption (for tablets); - fructose intolerance (for drops); - children under 4 years of age (for tablets); - hypersensitivity to sodium picosulfate or other components of the drug. Use during pregnancy and lactation for long periods There was no experience of using the drug with adverse events during pregnancy. However, due to the lack of research, the use of Guttalax® during pregnancy is recommended only if the expected benefit to the mother outweighs the potential risk to the fetus. Taking the drug in the first trimester of pregnancy is contraindicated. In the second and third trimesters of pregnancy, the drug can only be taken as prescribed by a doctor. The active metabolite and its glucuronides are not excreted in breast milk. Thus, the drug can be used during breastfeeding. Studies on the effect of the drug on fertility have not been conducted. In preclinical studies, no teratogenic effects on reproduction were identified.
Overdose
Symptoms When using the drug in high doses, diarrhea, dehydration, decreased blood pressure, water and electrolyte imbalance, hypokalemia, and convulsions are possible. In addition, there are reports of cases of ischemia of the muscles of the large intestine associated with taking the drug in doses significantly higher than those recommended for the usual treatment of constipation. The drug Guttalax®, like other laxatives, in chronic overdose can lead to chronic diarrhea, abdominal pain, hypokalemia, secondary hyperaldosteronism, urolithiasis. Due to chronic laxative abuse, renal tubular damage, metabolic alkalosis, and muscle weakness associated with hypokalemia may develop. Treatment To reduce the absorption of the drug after oral administration, vomiting or gastric lavage can be performed. Fluid replacement and correction of electrolyte balance may be required, as well as antispasmodics.
special instructions
You should not use the drug daily without consulting a doctor for more than 10 days. Long-term use of the drug in high doses can lead to fluid loss, electrolyte imbalance, and hypokalemia. Dizziness and fainting were observed in patients taking Guttalax®. The analysis showed that these cases are associated with syncope during defecation (or syncope caused by straining during defecation) or with a vasovagal response to abdominal pain, which may be due to constipation, and is not necessarily related to the drug. Per tablet (5 mg) contains 67.5 mg of lactose. The maximum recommended daily dose for the treatment of adults and children over 10 years of age and for children 4-10 years of age contains 135.0 mg and 67.5 mg of lactose, respectively. 1 ml of drops contains 0.45 mg of sorbitol. The maximum recommended daily dose for the treatment of adults and children 4-10 years old contains 0.6 g and 0.3 g of sorbitol, respectively. Use in pediatrics Children over 4 years of age should take the drug only as prescribed by a doctor. The drug in the form of drops has no taste, so children can be added to food. Effect on the ability to drive vehicles and operate machinery. Special clinical studies of the effect of the drug on the ability to drive vehicles or operate machinery have not been conducted. Despite this, patients should be advised that when using the drug, dizziness and/or fainting may occur due to a vasovagal reaction (i.e. during intestinal spasm). If intestinal spasms occur, patients should avoid potentially hazardous activities, incl. driving vehicles or operating machinery.
Overdose of the drug Guttalax, symptoms and treatment
Manifested by diarrhea, intestinal colic, water and electrolyte imbalance, in particular hypokalemia. There have been reports of ischemia of the colon mucosa when used in doses significantly higher than recommended. It has been reported that laxatives with long-term abuse have led to the development of chronic diarrhea and abdominal pain, hypokalemia, secondary hyperaldosteronism, possible renal tubular damage and the development of alkalosis, muscle weakness as a result of hypokalemia. In case of overdose, gastric lavage is carried out, water and electrolyte imbalances are corrected, which is especially important in the elderly and adolescents. Antispasmodics are used according to indications.
A country
Italy
The country of production may vary depending on the batch of goods. Please check with the operator for detailed information when confirming your order.
Compound
Bottle 15 ml
Sodium picosulfate monohydrate 0.75 g per 100 ml. Excipients: sodium benzoate - 0.2 g, sodium citrate dihydrate - 0.15 g, liquid sorbitol (non-crystallizing) - 64.37 g, citric acid monohydrate - 0.14 g, purified water - 49.89 g. Drops for oral administration in the form of clear, colorless to yellowish or slightly yellowish-brown, slightly viscous solution.
pharmachologic effect
The active substance, sodium picosulfate, is a laxative of the triarylmethane group. As a local laxative, sodium picosulfate, after bacterial breakdown in the large intestine, has a stimulating effect on the mucous membrane of the large intestine, increasing peristalsis, promoting the accumulation of water and electrolytes in the large intestine. This leads to stimulation of the act of defecation, reduction of evacuation time and softening of the stool. Sodium picosulfate, being a laxative acting at the level of the colon, stimulates the natural process of evacuation of contents from the lower gastrointestinal tract. Therefore, the drug does not affect the digestion or absorption of high-calorie foods or essential nutrients in the small intestine.
Indications for use
As a laxative in the following cases: - constipation caused by atony and hypotension of the colon (including in old age, in bedridden patients, after operations, after childbirth and during lactation); - constipation caused by taking medications; - to regulate stool in case of hemorrhoids, proctitis, anal fissures (to soften the consistency of stool); - gallbladder diseases, irritable bowel syndrome with a predominance of constipation; - constipation caused by intestinal dysbiosis and diet disorders.
Side effects
From the digestive system: discomfort, nausea, vomiting, cramps and pain in the abdomen, diarrhea. From the nervous system: dizziness and fainting. Dizziness and fainting that occur after taking the drug appear to be associated with a vasovagal response (eg, straining during bowel movements, abdominal cramps). From the immune system: hypersensitivity reactions, including angioedema, skin rash, itching.
Contraindications
- intestinal obstruction;
— obstructive intestinal diseases; - acute diseases of the abdominal organs or severe abdominal pain, which may be accompanied by nausea, vomiting, fever (including appendicitis); - acute inflammatory bowel diseases; - severe dehydration; - lactase deficiency, lactose intolerance, glucose-galactose malabsorption (for tablets); — fructose intolerance (for drops); - children under 4 years of age (for tablets); - hypersensitivity to sodium picosulfate or other components of the drug. Use during pregnancy and breastfeeding During long-term experience with the use of the drug, no adverse events during pregnancy were identified. However, due to the lack of research, the use of Guttalax® during pregnancy is recommended only when the expected benefit to the mother outweighs the potential risk to the fetus. Taking the drug in the first trimester of pregnancy is contraindicated. In the second and third trimesters of pregnancy, the drug can only be taken as prescribed by a doctor. The active metabolite and its glucuronides are not excreted in breast milk. Thus, the drug can be used during breastfeeding. Studies on the effect of the drug on fertility have not been conducted. In preclinical studies, no teratogenic effects on reproduction were identified.
Use in children Children under 4 years of age should not use the drug in tablet form; it is recommended to prescribe the drug in the form of drops.
Mode of application
The drug is taken orally. The tablets must be taken with a sufficient amount of liquid. The drops do not have to be dissolved in liquid. To obtain a laxative effect in the morning, you should take the drug the night before. Adults and children over 10 years of age are prescribed 1-2 tablets. or 10-20 drops (5-10 mg)/day. Children aged 4-10 years are prescribed 1/2-1 tablet. or 5-10 drops (2.5-5 mg)/day. Children under 4 years of age should not use the drug in tablet form; it is recommended to prescribe the drug in the form of drops. The recommended dose for children under 4 years of age is 0.25 mg/kg body weight/day. This corresponds to 1 drop of the drug (0.5 mg sodium picosulfate) per 2 kg of body weight/day. It is recommended to start treatment with a lower dose. In order to achieve regular bowel movements, the dose can be increased to the maximum recommended. The maximum recommended daily dose should not be exceeded.
special instructions
You should not use the drug daily without consulting a doctor for more than 10 days. Long-term use of the drug in high doses can lead to fluid loss, electrolyte imbalance, and hypokalemia. Dizziness and fainting have occurred in patients taking Guttalax®. The analysis showed that these cases are associated with syncope with defecation (or syncope caused by straining to defecate) or with a vasovagal response to abdominal pain, which may be due to constipation, and is not necessarily related to the drug. One tablet (5 mg) contains 67.5 mg of lactose. The maximum recommended daily dose for the treatment of adults and children over 10 years of age and for children 4-10 years of age contains 135.0 mg and 67.5 mg of lactose, respectively. 1 ml of drops contains 0.45 mg of sorbitol. The maximum recommended daily dose for the treatment of adults and children 4-10 years old contains 0.6 g and 0.3 g of sorbitol, respectively. Use in pediatrics Children over 4 years of age should take the drug only as prescribed by a doctor. The drug in the form of drops has no taste, so it can be added to children's food. Effect on the ability to drive vehicles and operate machinery. No special clinical studies have been conducted on the effect of the drug on the ability to drive vehicles or operate machinery. Despite this, patients should be advised that when using the drug, dizziness and/or fainting may occur due to a vasovagal reaction (i.e. during intestinal spasm). If intestinal spasms occur, patients should avoid potentially hazardous activities, incl. driving vehicles or operating machinery.
Overdose
Symptoms When using the drug in high doses, diarrhea, dehydration, decreased blood pressure, water and electrolyte imbalance, hypokalemia, and convulsions are possible. In addition, there are reports of cases of ischemia of the muscles of the large intestine associated with taking the drug in doses significantly higher than those recommended for the usual treatment of constipation. The drug Guttalax®, like other laxatives, in chronic overdose can lead to chronic diarrhea, abdominal pain, hypokalemia, secondary hyperaldosteronism, and urolithiasis. Renal tubular damage, metabolic alkalosis, and muscle weakness associated with hypokalemia may develop due to chronic laxative abuse. Treatment To reduce the absorption of the drug after oral administration, you can induce vomiting or perform gastric lavage. Fluid replacement and correction of electrolyte balance may be required, as well as antispasmodics.
Interaction with other drugs
Diuretics or corticosteroids increase the risk of electrolyte imbalance (hypokalemia) when using the drug in high doses. Electrolyte imbalance may increase sensitivity to cardiac glycosides. The combined use of the drug and antibiotics may reduce the laxative effect of the drug.