Compound
| Gelatin capsules | 1 caps. |
| active substance: | |
| standardized lyophilisate of bacterial lysates (Haemophilus influenzae, Streptococcus pneumoniae, Streptococcus viridans, Streptococcus pyogenes, Klebsiella pneumoniae, Klebsiella ozaenae, Staphylococcus aureus, Moraxella catarrhalis) | 7 mg |
| (as part of lyophilisate OM-85 - 40 mg) | |
| excipients: anhydrous propyl gallate (E310) - 0.084 mg; sodium glutamate - 3.03 mg; mannitol - up to 40 mg; pregelatinized starch - 110 mg; magnesium stearate - 3 mg; mannitol - up to 200 mg | |
| capsule shell: indigo carmine dye (indigotine) (E132) - 0.03 mg; titanium dioxide (E171) - 1 mg; gelatin - up to 50 mg |
Pharmacodynamics
Immunostimulating drug of bacterial origin. Causes an immune response in the mucous membrane of the digestive tract. The effect is especially pronounced in Peyer's patches (PB) of the small intestine. Antigen-presenting cells (APC) in the PB are activated by the drug and subsequently stimulate cells responsible for specific immunity.
When using the drug, an increase in the number of circulating B-lymphocytes is observed.
Following stimulation of B lymphocytes, there is an increase in the production of polyclonal antibodies, especially serum IgG and IgA secreted from the respiratory mucosa and saliva.
These antibodies are the first line of defense against a range of infectious agents (viruses and bacteria).
The drug has a powerful stimulating effect on most types of leukocytes, as evidenced by an increase in the number of myeloid and lymphoid cells, as well as a selective increase in the expression of receptors on their surface.
Taken together, these data confirm that the drug triggers biological reactions that enhance the body's immune defense against infection.
Clinically, Broncho-Vaxom® adult reduces the frequency of acute respiratory tract infections, shortens the duration of their course, reduces the likelihood of exacerbations of chronic bronchitis, and also increases the body's resistance to respiratory system infections. This reduces the need to use other drugs, especially antibiotics.
Broncho-Vaxom capsules 3.5 mg No. 10
Compound
Active ingredients: standardized lyophilisate of bacterial lysates: Haemophilus influenzae, Streptococcus pneumoniae, Streptococcus pyogenes, Streptococcus viridans, Klebsiella pneumoniae, Klebsiella ozaenae, Staphylococcus aureus, Moraxella catarrhalis 3.5 mg.
Excipients: anhydrous propyl gallate (E310) - 0.042 mg, sodium glutamate - 1.515 mg, mannitol - up to 20 mg, pregelatinized starch - 110 mg, magnesium stearate - 3 mg, mannitol - up to 200 mg. Composition of the capsule shell: indigo carmine (indigotine) dye (E132) - 0.01 mg, titanium dioxide (E171) - 1 mg, gelatin - up to 50 mg.
Pharmacokinetics
Data on the pharmacokinetics of the drug Broncho-Vaxom® for children are not provided.
Indications for use
Children aged 6 months to 12 years:
- prevention of recurrent respiratory tract infections and exacerbations of chronic bronchitis;
- treatment of acute respiratory tract infections (as part of complex therapy).
Contraindications
Hypersensitivity to the components of the drug.
Directions for use and doses
Broncho-Vaxom® for children is prescribed to children aged 6 months to 12 years.
For the treatment of acute and exacerbation of chronic respiratory infections: 1 capsule of Broncho-Vaxom® for children daily in the morning on an empty stomach until symptoms disappear, but for at least 10 days. If antibiotic therapy is necessary, Broncho-Vaxom® for children should be taken in combination with an antibiotic from the beginning of treatment.
The duration of treatment should be determined by the doctor based on the patient’s health condition.
For the prevention of exacerbations and maintenance therapy: 1 capsule of Broncho-Vaxom® for children daily in the morning on an empty stomach. The course includes 3 cycles, each of which consists of taking 1 capsule daily for 10 days, the interval between cycles is 20 days.
For children under 3 years of age or if the child has difficulty swallowing the capsule, it should be opened and the contents mixed with a drink (water, fruit juice, milk).
Storage conditions
The drug should be stored out of the reach of children at a temperature of 15° to 25°C.
Best before date
5 years. Do not use after the expiration date stated on the packaging.
special instructions
Broncho-Vaxom® for children should not be used in children under 6 months of age.
To avoid overdose, you should not use the drug from capsules intended for adults (Broncho-Vaxom® adult capsules 7 mg).
Components of the drug may cause hypersensitivity reactions. In case of persistent gastrointestinal disorders, skin reactions, respiratory disorders or other symptoms of intolerance to the drug, treatment should be discontinued, because These symptoms are a manifestation of allergic reactions.
Description
Immunostimulating agent.
Use in children
Broncho-Vaxom® for children should not be used in children under 6 months of age.
Pharmacodynamics
Immunostimulating drug of bacterial origin. Causes an immune response in the mucous membrane of the digestive tract. The effect is most pronounced in Peyer's patches (PB) of the small intestine. Antigen-presenting cells (APC) in the PB are activated by the drug and subsequently stimulate cells responsible for specific immunity.
When using the drug, an increase in the number of circulating B-lymphocytes is observed. Following stimulation of B lymphocytes, there is an increase in the production of polyclonal antibodies, especially serum IgG and IgA secreted from the respiratory mucosa and saliva. These antibodies are the first line of defense against a range of infectious agents (viruses and bacteria).
The drug has a powerful stimulating effect on most types of leukocytes, as evidenced by an increase in the number of myeloid and lymphoid cells, as well as a selective increase in the expression of receptors on their surface.
Taken together, these data confirm that the drug triggers biological reactions that enhance the body's immune defense against infection.
Clinically, Broncho-Vaxom® for children reduces the frequency of acute respiratory tract infections, shortens the duration of their course, reduces the likelihood of exacerbation of chronic bronchitis, and also increases the body's resistance to respiratory system infections. This reduces the need to use other medications, especially antibiotics.
Side effects
Broncho-Vaxom® for children is usually well tolerated. Most adverse reactions fall into the general category of moderate to moderately severe. The most common side effects of the drug are skin reactions, disorders of the digestive tract and respiratory system.
In each particular category, side effects are grouped by system-organ class and presented in descending order of frequency: very often (≥1/10); often (from 1/100 to 1/10); uncommon (from 1/1000 to 1/100); rare (from 1/10,000 to 1/1000); very rare (<1/10,000); unknown (cannot be estimated based on available data).
From the digestive system: often - diarrhea, abdominal pain; unknown - vomiting, nausea.
From the nervous system: unknown - headache.
From the respiratory system: often - cough.
Dermatological reactions: often - rash.
Allergic reactions: uncommon - hypersensitivity reactions (erythematous rash, generalized rash, erythema, eyelid swelling, facial swelling, peripheral edema, facial swelling, itching, generalized itching, shortness of breath); unknown - urticaria, Quincke's edema.
Other: unknown - fever, fatigue.
Interaction
Broncho-Vaxom® for children can be used simultaneously with other medications for the treatment of acute and chronic respiratory diseases.
The interaction of the drug Broncho-Vaxom® for children with other drugs has not yet been established.
Overdose
There are no data on cases of overdose. The nature of the drug Broncho-Vaxom® for children and the results of studying its toxicity indicate that an overdose is unlikely.
Impact on the ability to drive vehicles and operate machinery
Broncho-Vaxom® for children does not affect the ability to drive vehicles or operate machinery.
Side effects
The immunostimulating drug Broncho-Vaxom® for adults is usually well tolerated. Most adverse reactions fall into the general category of moderate to moderately severe. The most common side effects of the drug are gastrointestinal upset, skin reactions, and respiratory disorders.
In each particular category, side effects are grouped by system-organ class and presented in descending order of frequency: very often (≥1/10); often (from ≥1/100 to <1/10); uncommon (from ≥1/1000 to <1/100); rare (from ≥1/10000 to <1/1000); very rare (<1/10000); unknown (cannot be estimated based on available data).
From the gastrointestinal tract: often - diarrhea, abdominal pain; unknown - vomiting, nausea.
From the nervous system: unknown - headache.
From the respiratory system, chest and mediastinal organs: often - cough.
From the skin and subcutaneous tissue: often - rash; unknown - urticaria, Quincke's edema.
General disorders and administration site disorders: unknown - fever, fatigue.
From the immune system: rarely - hypersensitivity (erythematous rash, generalized rash, erythema, eyelid swelling, facial swelling, peripheral edema, facial swelling, itching, generalized itching, shortness of breath).
Directions for use and doses
Inside.
Treatment: 1 capsule. Broncho-Vaxom® for adults daily in the morning on an empty stomach until symptoms disappear, but for at least 10 days. If antibiotic therapy with Broncho-Vaxom® is necessary, adults should take it in combination with an antibiotic from the beginning of treatment.
The duration of treatment or the appointment of a second course of therapy should be determined by the doctor, based on the patient’s health condition.
Prevention of exacerbations and maintenance therapy: 1 caps. Broncho-Vaxom® for adults daily in the morning on an empty stomach. The course includes 3 cycles, each of which consists of a daily dose of 1 capsule. for 10 days, the interval between cycles is 20 days.
If the capsule is difficult to swallow, it should be opened and the contents mixed with something to drink (including water, fruit juice, milk).
BRONCHO-VAXOM CHILDREN (capsules)
prescribed us to take “Broncho-Vaxom for children” according to the scheme: take ten days in the morning on an empty stomach, twenty days break from taking the medicine.
Thus, it is necessary to undergo a course of treatment, which includes three courses. Therefore, it is more profitable to buy a package that contains 30 capsules. I bought the 10-capsule form of the drug because I was afraid of allergic reactions in my daughter. One such package cost me 540 rubles. So far we have completed the first course of Pharma's immunostimulating drug "Broncho-Vaxom for children." During all ten days of taking the drug, my daughter went to kindergarten; on the third day of her stay in the child care center, the child began to have a slight runny nose. I continued to give the child “Broncho-Vaxom for children.” Ten days after finishing taking the medicine, my daughter woke up with a temperature of thirty-nine and a slight sore throat. The pediatrician prescribed antiviral treatment. Three days later, my daughter’s voice became hoarse, the doctor prescribed Erespal. This drug is prescribed for diseases of the upper and lower respiratory tract. I followed all the doctor’s orders, gave my daughter Erespal as prescribed by the doctor, gave her plenty to drink, maintained the temperature in the apartment, and maintained the required level of humidity. Every three days the child's breathing was observed and listened to by a pediatrician. Three days later, the child’s temperature rose to thirty-nine, the doctor heard wheezing and prescribed antibiotics. My daughter was diagnosed with bronchitis.
I literally described the course of the child’s illness in order to paint a general picture of the child’s well-being after taking the first course of “Broncho-Vax for children.” Of course, the manufacturer notes that to obtain the effect, it is necessary to undergo a course of treatment consisting of three cycles. However, disappointment does not leave me. The first course of the immunostimulating drug Pharma “Broncho-Vaxom for children” turned out to be completely useless. My daughter developed not just a cough, but bronchitis. Moreover, this is the first bronchitis in three years, which is how old my daughter is now. That is, for there to be any effect, it takes time and a considerable amount of money.
Now the time has come for the second course of this drug, I purchased it. My daughter is still taking antibiotics, and what do I see in the instructions for use of the medicine? The manufacturer indicates that the drug must be taken in combination with an antibiotic from the beginning of treatment. And the time has come for us to take the second course at a time when there are a few days left before the end of taking the antibiotic.
At the moment, after the first course of the immunostimulating drug Pharma Broncho-Vax, I am not satisfied with it. Perhaps the second and third course of this drug will produce results and the child will not suffer from acute respiratory tract infections. Now I am inclined to believe that this drug is another way of pumping money out of parents, money that can be spent on fruits or other nutritious, healthy food for the child.
I give the drug three stars only because we did not complete the full course of treatment. At the moment the drug is disappointing!
special instructions
Components of the drug may cause hypersensitivity reactions.
In case of persistent gastrointestinal disorders, skin reactions, respiratory problems or other symptoms of intolerance to the drug, treatment should be discontinued, because These symptoms are a manifestation of an allergic reaction.
Impact on the ability to drive vehicles and machinery. Broncho-Vaxom® adult does not affect the ability to drive vehicles and machines.
Bacterial lysates. Broncho-Vaxom
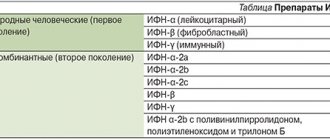
Preparations containing bacterial lysates have attracted the interest of many specialists in recent years; their main use is primarily associated with the prevention of infections of the nasopharynx and respiratory tract. Considering the immunotropic effect and the lack of formation of stable protective immunity, it is more correct to call this group of drugs bacterial immunomodulators. Based on the composition and assessment of the features of the mechanism of action, they can be divided into groups. Bacterial immunomodulators: I. Lysates of microorganisms - synthetic preparations: Broncho-Vaxom Broncho-munal Ribomunil VP-4 (multicomponent vaccine) Biostim IRS-19 Imudon Solkourovak Ruzam Solkotrichovak Salmozan Prodigiozan Pyrogenal Immuforce II Systemic action: Broncho-Vaxom Broncho-munal Ribomunil (tablets form) Biostim VP-4 Likopid (tablet form 10 mg) Ruzam Immufors III Mainly topical action: IRS-19 Imudon Lykopid (tablet form 1 mg) Many drugs (prodigiosan, pyrogenal, salmosan) have only historical significance and are not currently used. Among the new bacterial lysates in clinical trials is the drug Immufors, which contains Shigella sonnei lipopolysaccharide, has low pyrogenicity and toxicity, has a targeted effect on the macrophage component of the immune system, and stimulates the cytotoxic and antigen-presenting activity of macrophages. Under the influence of immuforce, the synthesis of cytokines such as interferon-g (IFN), tumor necrosis factor-a (TNF), and interleukin-6 (IL-6) increases. The effect on the functional activity of macrophages is especially clearly manifested with an initial decrease in this indicator. Broncho-Vaxom (pharmaceutical, Switzerland) includes lyophilized extracts of 8 pathogens: Haemophilus influenzae, Diplococcus pneumonia, Streptococcus viridans, Streptococcus pyogens, Klebsiella pneumoniae, Klebsiella ozaena, Staphylococcus aureus, Neisseria (Branhamella) catarrhalis. Broncho-Vaxom is approved for use from 6 months and is available in dosages of 7 mg and 3.5 mg. The drug is usually well tolerated, the frequency of mild adverse reactions is 3–4%. In children, if necessary, the capsule can be opened and the contents dissolved in juice or water. Adverse reactions manifest themselves in the form of gastrointestinal disorders (nausea, abdominal pain, vomiting), skin reactions (including erythema and itching), respiratory system disorders (cough, shortness of breath), as well as hunger pain and fatigue. In case of ongoing gastrointestinal disorders, skin reactions and respiratory manifestations, treatment should be discontinued. Prescription regimen for Broncho-Vaxom: in adults and children over 12 years of age, 7 mg capsule, in the morning on an empty stomach, 10 days a month – 3 months. In children from 6 months to 12 years, the drug is prescribed in capsules of 3.5 mg. Broncho-Vaxom stimulates the secretion of prostaglandin E2 and IL-1 by alveolar macrophages, increases the activity of macrophages against infectious agents and tumor cells, enhances the secretion of aTNF and IL-2, activates natural killer cells (NK cells), enhances the synthesis of IgA, IL-8, aTNF , neutrophil-activating factor (NAF) [14,16]. The drug stimulates B cells, increases the level of IgA, secretory IgA on mucous membranes, in saliva, bronchoalveolar lavage fluid, increases the functional and metabolic activity of macrophages, including alveolar macrophages [14,16,18]. Bacterial lysates can be prescribed during the acute period of the disease and for prophylactic purposes. In the acute period of respiratory infections, the prescription of drugs is more effective in combination with etiotropic therapy. In acute respiratory viral infections (ARVI), inflammation is localized in various parts of the upper respiratory tract, which can lead to the development of nasopharyngitis, pharyngitis-laryngitis, laryngo-tracheo-bronchitis, etc. A predisposing factor is a decrease in local immunity. When observing 45 patients with acute pharyngitis or exacerbation of chronic pharyngitis aged from 19 to 65 years, 15 of them received Broncho-Vaxom as monotherapy, 1 capsule for 10 days. 15 patients received Broncho-Vaxom in combination with local administration of Povidone iodine (an antiseptic and disinfectant). 15 patients received topical Povidone iodine. The best effect was obtained in patients with acute pharyngitis who received combination therapy with Broncho-Vaxom and Povidone-iodine [10]. 25–50% of children with respiratory infections receive various antibiotics, as pediatricians are afraid to miss the activation of bacterial flora after a viral infection [3]. According to our data, in frequently ill children with acute respiratory infections, the administration of bacterial lysates in combination with antibacterial therapy is more effective when culturing two or more bacterial pathogens from the throat and nasopharynx. When Broncho-Vaxom was prescribed in the acute period of respiratory infections to children for 10 days in combination with antibiotics, the effectiveness of complex use and a reduction in the need for other drugs and antibiotics was noted [2]. According to our data, when prescribing systemic bacterial lysates in a group of frequently and long-term ill children (CHI) with foci of chronic infection (chronic tonsillitis, chronic pharyngitis, adenoiditis) and a frequency of acute respiratory infections 6 or more times a year, outside the period of exacerbation, activation of T– cells (CD3+HLA-DR+ cells), an increase in the number of CD3+, CD4+ cells, T-killer cells (CD3+CD16+ cells), synthesis of a and g-IFN by cells, levels of serum IgG, IgA and IgA in saliva. The geometric mean titer of IgG-AT to bacterial pathogens (Staphylococcus spp., Streptococcus spp., Klebsiella spp., Neisseria spp.) decreases. The IgG-AT titer to these pathogens is increased in the ChBD, which is associated with the persistence of the infection and is confirmed by cultures of the throat flora. A decrease in IgG antibody titer may be associated with the destruction of infectious agents with the help of killer T cells and the synthesis of IFN. The effect of one course of systemic drugs lasts for 6 months, and one course of topical drugs lasts for 3 months [4,5,6,7]. The FCD examination showed a more pronounced persistence of infection in this group of patients compared to children with a frequency of acute respiratory infections less than 6 times a year (Table 1). A monoculture was isolated from 40% of children, two or more pathogens were isolated from 46.6% of children, Candida albicans was isolated from 25% of children, and a combined flora was isolated from 25% of the examined children. With a decrease in the frequency of acute respiratory infections, the frequency and range of sown microorganisms decreases. A decrease in the sensitivity of pathogens to various antibiotics leads to the persistence of resistant strains in BBD (Table 2). In 13.3% of the patients, a concomitant recurrent herpetic infection was diagnosed (the number of relapses was 5–9 times a year). Antibodies to Mycoplasma pneumonia were detected in 50% of the patients (IgM AT titer 1:8–1:32, IgG AT titer 1:16–1:64). In 30% of children, antibodies to Chlamydia pneumonia were determined (IgM AT titer 1:8–1: 64; IgG AT titer 1:16–1:128. In 40–20% of children, the infection was confirmed by PCR diagnostics (Mycoplasma and Chlamydia pneumonia, respectively). In 30% of children, the infection was mixed. The presented data indicate persistence of infection in the nasopharynx, while confirmation of infection with Streptococcus haemoliticus-b, Chlamydia and Mycoplasma pneumonia is an indication for antibiotic therapy [8]. Persistence is primarily bacterial infections in ChBD confirms the validity of prescribing bacterial lysates. According to Kubylinskaya I.A., risk factors for the occurrence of respiratory infections in children are age (usually 1–5 years), time of year (usually in winter), the number of children in an organized group. Broncho-Vaksom prescribed for the prevention of upper respiratory tract infections in children attending preschool and school institutions, children at risk (frequently ill), children with immunodeficiency conditions. The drug had a preventive effect. A repeated course of the drug was carried out 6 months from the start of treatment. The Broncho-Vaxom effect was manifested in a decrease in the frequency and duration of upper respiratory tract infections, a twofold decrease in the number of exacerbations of chronic diseases of the ENT organs, a decrease in the frequency and severity of infections, and the need for antibiotics [3]. In a double-blind, randomized, placebo-controlled study of 220 children with repeated respiratory infections, the administration of Broncho-Vaxom led to a significant reduction in the incidence of infections [17]. A comparative assessment of the clinical and immunological effectiveness of Broncho-Vaxom was carried out in children aged 2–7 years with repeated bronchitis, nasopharyngotracheitis, chronic adenoiditis in the conditions of sanatorium rehabilitation treatment. 19 children received sanatorium treatment, which included diet therapy, regimen, physical therapy, massage, multivitamins, and nasopharyngeal sanitation (furacilin, calendula, eucalyptus, sea salt solution). 32 children received Broncho-Vaxom according to the standard regimen. After 5 months, the number of respiratory infections in children receiving Broncho-Vaxom decreased by 2 times, and the average duration of episodes decreased by 2.4 times; The frequency of recurrent bronchitis decreased most significantly – by 8 times [9]. In adult patients with chronic bronchitis, after prescribing Broncho-Vaxom for prophylactic purposes, the frequency and severity of exacerbations, the need for antibiotics, and the inoculation of bacterial flora from sputum significantly decreased [11,13]. In patients with repeated respiratory infections and hypogammaglobulinemia after treatment with Broncho-Vaxom, an increase in the level of serum IgG, A was noted [15]. In patients with common variable immunological deficiency (CVID), the level of IgA increased after taking Broncho-Vaxom [12]. At present, Broncho-Vaxom cannot be the drug of choice in the treatment of hypogammaglobulinemia or one of the nosological forms of primary immunodeficiency - CVID, since in the treatment of these diseases, immunoglobulin preparations are more often used to increase the level of immunoglobulins. It should be noted that the administration of one course of bacterial lysates does not lead to a complete cessation of episodes of respiratory infections. The preventive effect when prescribing drugs outside the acute period can be assessed by the frequency of acute respiratory infections episodes and the dynamics of immunological indicators, in particular, for systemic drugs - by the dynamics of systemic immunity indicators, and for topical drugs - by indicators of local immunity [1,4,5, 8.6]. Thus, the use of bacterial lysates (Broncho-Vaxom) reduces the frequency of exacerbations of diseases of the bronchopulmonary system, reduces the risk of rapid progression of the inflammatory process in the lungs, reduces the frequency of exacerbations of chronic diseases of the ENT organs (rhinitis, sinusitis, laryngitis, tracheitis), reduces the frequency of reinfection ARVI. The use of Broncho-Vaxom reduces the need for antibiotics, which avoids the immunosuppressive effects of antibiotics, the emergence of resistant strains and the development of other complications of antibiotic therapy.
Literature. 1. Bogomilsky M.R., Markova T.P., Garashchenko T.I., Chuvirov D.G. and others. Clinical and immunological rationale for the use of the topical bacterial immunocorrector IRS19 for the prevention of upper respiratory tract diseases in children. Children's Doctor, 2000, No. 5, pp. 4–7. 2. Karaulov A.V., Sokurenko S.I., Barmotin G.V. Principles of immunoprophylaxis and immunotherapy of recurrent respiratory diseases. MMA im. Sechenov, clinic of the Ministry of Foreign Affairs of the Russian Federation. M., 1999. 3. Kubylinskaya I.A. Pharmacotherapy: Use of Broncho-Vaxom in the prevention and treatment of acute and chronic diseases of the ENT organs in children. Abstract, 2006, No. 15 (420). 4. Markova T.P., Chuvirov D.G., Garashchenko T.I. The mechanism of action and effectiveness of broncho-munal in a group of long-term and frequently ill children. Immunology, 1999, no. 6, pp. 56–59. 5. Markova T.P., Chuvirov D.G. The use of topical immunomodulators in a group of long-term and frequently ill children. In the book: Immunocorrection in Pediatrics, ed. M.V. Kostinova. M., 2001, pp. 91–99. 6. Markova T.P., Chuvirov D.G. Bacterial immunomodulators. RMJ, 2001, 9 (16–17), pp. 703–6. 7. Markova T.P., Chuvirov D.G. Clinical and immunological rationale for the use of imudon in long-term and frequently ill children. Children's Doctor, 2001, No. 7, pp. 2–4. 8. Markova T.P., Chuvirov D.G. Long-term and frequently ill children. RMJ, 2002, vol. 10, no. 3 (147), pp. 125–129. 9. Mizernitsky Yu.L., Melnikova I.M., Marushkov V.I. Immunocorrection in complex therapy of respiratory diseases. Pulmonology, supplement to Konsilium Medicum, 2005, vol. 7, no. 16. 10. Ovchinnikov A.Yu., Svistushkin V.M., Nikiforova G.N. Treatment of tonsillopharyngitis, as one of the clinical manifestations of ARVI. Proceedings of the IV All-Russian scientific and practical conference “Reliability and reliability of scientific information in otorhinolaryngology” (November 9–10, 2005, Moscow). 11. Li J., Zheng JP., Yuan JP. et al. Clin.Med.J.Engl., 2004, 21 (14), P. 1053–1068. 12. Litzman J., Lokaj J., Gerylovova A. Allerg. Immunol. (Paris), 1996, 28 (3), pp. 81–85. 13. Marialigeti T., Szekely E., Kereky E. Ther. Hung., 1989, 37 (1), pp. 28–31. 14. Maul J. Stimulation of immunoprotective mechanisms by OM–85 BV. Respiration, 1994, No. 61 (Suppl. 1), P.8–15. 15. Quezada A., Maggi L., Perez M., Rodriguez JJ Investig. Allergol. Clin. Immunol., 1999, 9 (3), P. 178–82. 16. Roth M., Keul R., Papakonstantinou E. et al. Characterization of intracellular signaling transduction and transcription factors involved in Broncho–Vaxom (OM–85 BV) – induced expression of interleukin-6 and interleukin-8 in human pulmonary fibroblasts. Eur.Respira. Rev., 1996, V.6, No. 38, P.171–175. 17. Schaad B., Mitterlein R., Goffin H. Chest, 2002, 122, P. 2042–49. 18. Zagar S., Lofler-Badzek D., Cvoriscec BJ. Broncho-Vaxom in children with rhinosinusitis. A double-blind clinical trial. Otorhinolaryngol. Relat. Spec., 1988, v.50, pp. 397–404.