Gynokaps
The pharmacological activity of the drug Ginocaps is due to the complex action of its components - metronidazole and miconazole nitrate. The drug has antifungal, antiprotozoal and antibacterial effects.
Metronidazole is an antimicrobial and antiprotozoal agent, a derivative of 5-nitroimidazole. The mechanism of action is the biochemical reduction of the 5-nitro group of metronidazole by intracellular transport proteins of anaerobic microorganisms and protozoa. The reduced 5-nitro group of metronidazole interacts with the DNA of microbial cells, inhibiting the synthesis of their nucleic acids, which leads to the death of bacteria. Active against Trichomonas vaginalis, Gagdnerella vaginalis, Giardia intestinalis, Entamoeba histolyca, Lamblia spp., as well as obligate anaerobes Bacteroides spp. (including Bacteroides fragilis, Bacteroides distasonis, Bacteroides ovatus, Bacteroides thetaiotaomicron, Bacteroides vulgatus), Fusobacterium spp., Veillonella spp., Prevotella (Prevotella bivia, Prevotella buccae, Prevotella disiens) and some gram-positive microorganisms (Eubacter spp. Clostridium spp., Peptococcus spp., Peptostreptococcus spp., Mobiluncus spp.). Aerobic microorganisms are insensitive to metronidazole.
Miconazole is an antifungal agent that inhibits the synthesis of ergosterol in the cell membrane. Active against dermatophytes, yeast and some other fungi. Shows antimicrobial activity against gram-positive microorganisms. Eliminates itching that usually accompanies fungal infections. When used intravaginally, it is active mainly against Candida albicans.
Pharmacokinetics
Suction
When administered intravaginally, metronidazole is absorbed into the systemic circulation. The bioavailability of metronidazole during intravaginal use of Ginocaps capsules is 20% of the administered dose.
Systemic absorption of miconazole nitrate with this route of administration is very low (approximately 1.4% of the dose).
Metabolism and excretion
Metronidazole is metabolized in the liver by hydroxylation, oxidation and glucuronidation. The activity of the main metabolite (2-oxymetronidazole) is 30% of the activity of the parent compound.
T1/2 of metronidazole is 6-11 hours. Excreted by the kidneys - 60-80% of the dose of the systemic drug (20% of this amount unchanged). The metabolite of metronidazole, 2-hydroxymetronidazole, colors urine red-brown due to the presence of a water-soluble pigment formed as a result of the metabolism of metronidazole. The intestines excrete 6-15% of the dose of the systemic drug.
Penetrates into breast milk and most tissues, passes through the blood-brain barrier and placenta.
Miconazole is metabolized in the liver to form inactive metabolites. Miconazole overcomes histohematic barriers poorly and practically does not penetrate into the CSF.
8 hours after using the drug, 90% of miconazole is still present in the vagina. Unchanged miconazole is not detectable in either plasma or urine.
Instructions for use
Gynocaps is used intravaginally and inserted deep into the vagina.
For trichomoniasis, 1 vaginal capsule is prescribed 1 time per day in the evening before bed for 10 days in combination with oral metronidazole.
For bacterial vaginosis, 1 vaginal capsule is prescribed 1 - 2 times a day for 10 days, if necessary, in combination with oral metronidazole.
For vulvovaginal candidiasis, 1 vaginal capsule is inserted deep into the vagina once a day for 10 days.
special instructions
During treatment with the drug and for at least one day after treatment, alcohol consumption is prohibited (disulfiram-like reactions may develop: spastic abdominal pain, nausea, vomiting, headache, sudden flushing of the face) and abstinence from sexual intercourse is recommended.
When treating trichomoniasis, it is advisable to simultaneously treat the sexual partner with metronidazole tablets for oral administration.
It is advisable to prescribe vaginal capsules in combination with oral forms of metronidazole.
If the drug is used together with metronidazole for oral administration, especially with a repeated course, monitoring of the peripheral blood picture is necessary (danger of leukopenia).
With caution: pregnancy (II-III trimesters), history of leukopenia.
Pregnancy and breastfeeding period
In the first trimester of pregnancy, the use of the drug is contraindicated.
Metronidazole penetrates the blood-placental barrier.
Tests were carried out on rats, in which it was determined that when a five-fold dose was administered to rats, there was no negative effect of the drug on either the fertility of adult rats or the fetus. When the usual dose recommended for human treatment was administered intraperitoneally to pregnant mice, an increase in fetotoxicity was noted, but when the same dose was administered orally, no abnormalities were found.
Despite this, no studies have been conducted in pregnant women.
A meta-analysis of studies with the administration of metronidazole in the first three months of pregnancy was conducted and concluded that there was no increase in negative effects on the fetus.
Despite this, metronidazole during pregnancy should only be prescribed after a careful assessment of the expected benefits and disadvantages of using the drug.
Lactation
Oral metronidazole is found in human milk at concentrations equal to plasma concentrations. It can give milk a bitter taste. To avoid a negative effect of the drug on the child, you must stop either breastfeeding during treatment and after stopping the course for another 1-2 days, or use the drug, depending on how important the course of therapy is for the mother.
Use of the drug in elderly patients
Use in elderly patients requires caution due to age-related changes in the pharmacokinetics of metronidazole.
Impact on the ability to drive a car, work with equipment
Does not affect.
Interaction with other drugs
The drug may enhance the effect of oral anticoagulant drugs. Prothrombin time may increase, so dose adjustment of oral anticoagulants is necessary.
Enzyme inducers (eg: phenytoin, phenobarbital) can accelerate the elimination of metronidazole, which will lead to a decrease in its plasma level while increasing the clearance of phenytoin.
Enzyme inhibitors (eg: cimetidine) may increase the half-life and reduce the clearance of metronidazole.
Concomitant use of alcohol causes reactions similar to disulfiram (cramping abdominal pain, nausea, vomiting, headache, redness of the skin).
Concomitant use with disulfiram is not permissible (additive effects, may cause a psychotic state, confusion).
The level of lithium in the blood may increase during the course of treatment with metronidazole, therefore, before starting to use Ginocaps, it is necessary to reduce the dose of lithium or stop taking it for the duration of treatment.
Concomitant administration of cyclosporine with metronidazole may lead to an increase in the plasma level of cyclosporine, which requires laboratory monitoring (determining the level of cyclosporine in the blood plasma).
Metronidazole reduces the clearance of 5-fluorouracil, which increases its toxicity.
The drug may interfere with laboratory tests of ALT, AST, lactate dehydrogenase, glucose hexokinase, and triglyceride levels.
Side effect
Local: rarely - irritation of the vaginal mucosa and hypersensitivity reactions.
Systemic effects may develop:
From the gastrointestinal tract: nausea, vomiting, loss of appetite, cramping pain in the lower abdomen, diarrhea, coated tongue, bitter, metallic taste in the mouth.
From the hematopoietic system: reversible neutropenia (leukopenia).
From the side of the central nervous system: peripheral neuropathy (feeling of numbness of the extremities), rarely and only with long-term use, headaches, convulsions, drowsiness, dizziness, loss of coordination, ataxia, confusion occur.
Allergic reactions: skin rash, urticaria, skin itching, erythema multiforme, angioedema and anaphylactic reaction were very rare.
Effect of the drug on the liver: increased activity of liver enzymes, cholestasis, and jaundice have been rarely reported.
Others: fever, darkening of urine (causes a metabolite of metronidazole, has no clinical significance).
If side effects occur, treatment with Ginocaps should be discontinued.
Overdose
It is used exclusively intravaginally. In case of accidental ingestion of a large dose, gastric lavage is indicated.
If symptoms of overdose appear (nausea, vomiting, ataxia), only symptomatic therapy is performed (gastric lavage, administration of activated charcoal, hemodialysis), since metronidazole does not have a specific antidote. Metronidazole and its metabolites are readily eliminated by hemodialysis.
»
Ginocaps BIFORT vaginal capsules 200mg/750mg No. 5x2
Name
Ginocaps biforte
Description
Soft gelatin capsules, ovoid in shape with a pointed end, with a seam, elastic, opaque, almost white or light beige in color.
Main active ingredient
Metronidazole+miconazole
Release form
Vaginal capsules. 5 capsules in a blister pack, 2 or 3 blister packs along with an insert in a pack.
Dosage
200 mg+750 mg
special instructions
During treatment with the drug and for at least three days after treatment, alcohol consumption is prohibited (disulfiram-like reactions may develop: spastic abdominal pain, nausea, vomiting, headache, sudden flushing of the face) and abstinence from sexual intercourse is recommended. When treating trichomoniasis, it is advisable to simultaneously treat the sexual partner with metronidazole tablets for oral administration. It is advisable to prescribe vaginal capsules in combination with oral forms of metronidazole. If the drug is used together with metronidazole for oral administration, especially with a repeated course, monitoring of the peripheral blood picture is necessary (danger of leukopenia). With caution: pregnancy (Il–Ill trimesters), history of leukopenia. During therapy, you should not use other vaginal products (tampons, douches, spermicides). The results may change when determining the level of liver enzymes, glucose (hexokinase method), theophylline and procainamide in the blood. High doses and long-term systemic use of metronidazole can cause peripheral neuropathy and epilepsy. Do not swallow or otherwise administer!
Indications for use
Ginocaps Biforte is used for local treatment of infectious diseases of the vagina: bacterial vaginosis; trichomoniasis; candidal vaginitis; mixed vaginal infection.
Directions for use and doses
Ginocaps Biforte is used intravaginally, 1 vaginal capsule is inserted deep into the vagina for 7-10 days at night. For recurrent vaginitis or vaginitis resistant to other treatment: 1 vaginal capsule for 14–15 days. For the treatment of trichomonas vaginitis, it is advisable to combine Ginocaps Biforte vaginal capsules with oral forms of metronidazole or other systemic trichomonasid drugs. At the same time, it is necessary to treat the sexual partner with metronidazole tablets or other systemic trichomonacid drugs. Children: Not recommended for children. Patients with renal/hepatic insufficiency: Renal insufficiency: the half-life of metronidazole does not change. No dose reduction is required. However, in severe cases requiring hemodialysis, dose adjustment is necessary. In cases of severe hepatic impairment, metronidazole clearance may be impaired. At high plasma levels of metronidazole, an increase in symptoms of encephalopathy may occur, so metronidazole should be used with caution in patients with hepatic encephalopathy. The daily dose in patients with hepatic encephalopathy should be reduced to 1/3.
Use during pregnancy and lactation
Ginocaps Biforte capsules can be used after the first trimester of pregnancy under medical supervision, provided that the expected benefit to the mother outweighs the potential risk to the fetus. Breastfeeding should be stopped during treatment, since metronidazole passes into breast milk. Breastfeeding can be resumed 24–48 hours after the end of treatment.
Precautionary measures
Use of the drug in elderly patients Use in elderly patients requires caution due to age-related changes in the pharmacokinetics of metronidazole. Impact on the ability to drive a car, work with equipment. No effect.
Interaction with other drugs
The drug may enhance the effect of oral anticoagulant drugs. Prothrombin time may increase, so dose adjustment of oral anticoagulants is necessary. Enzyme inducers (eg: phenytoin, phenobarbital) can accelerate the elimination of metronidazole, which will lead to a decrease in its plasma level while increasing the clearance of phenytoin. Enzyme inhibitors (eg: cimetidine) may increase the half-life and reduce the clearance of metronidazole. Concomitant use of alcohol causes reactions similar to disulfiram (cramping abdominal pain, nausea, vomiting, headache, redness of the skin). Concomitant use with disulfiram is unacceptable (additive effects, can cause a psychotic state, confusion). The level of lithium in the blood may increase during the course of treatment with metronidazole, therefore, before starting to use Ginocaps Biforte, it is necessary to reduce the dose of lithium or stop taking it for the duration of treatment. Concomitant administration of cyclosporine with metronidazole may lead to an increase in the plasma level of cyclosporine, which requires laboratory monitoring (determining the level of cyclosporine in the blood plasma). Metronidazole reduces the clearance of 5-fluorouracil, which increases its toxicity. Metronidazole and miconazole inhibit the metabolism of astemizole and terfenadine and increase their plasma concentrations. The drug may interfere with laboratory tests of ALT, AST, lactate dehydrogenase, glucose hexokinase, and triglyceride levels.
Contraindications
severe liver dysfunction; epilepsy; I trimester of pregnancy, lactation period; hypersensitivity to the components of the drug; porphyria; in patients who drink alcohol during treatment or within 3 days after the end of treatment; in patients taking disulfiram during treatment or planning to use it within 2 weeks after completion of treatment.
Compound
One capsule contains: metronidazole - 750 mg, miconazole nitrate - 200 mg. Excipients - beeswax, migliol 812 N. Composition of the gelatin capsule shell: gelatin, glycerin, purified water, methyl parahydroxybenzoate E-218, propyl parahydroxybenzoate E-216, titanium dioxide E-171.
Overdose
There are no data regarding overdose with intravaginal use of metronidazole. After insertion into the vagina, metronidazole can be absorbed in quantities sufficient to cause systemic effects. Symptoms: nausea, vomiting, abdominal pain, diarrhea, generalized itching, metallic taste in the mouth, movement disorders (ataxia), dizziness, paresthenia, convulsions, peripheral neuropathy (including after prolonged use in high doses), leukopenia, darkening of urine. Treatment: in case of accidental ingestion, gastric lavage can be performed if necessary. There is no special antidote. Symptomatic and supportive therapy is recommended.
Side effect
Local: rarely - irritation of the vaginal mucosa (burning, itching). If severe irritation occurs, treatment should be discontinued. Systemic effects may develop: From the gastrointestinal tract: nausea, vomiting, loss of appetite, cramping pain in the lower abdomen, diarrhea, coated tongue, bitter, metallic taste in the mouth. From the hematopoietic system: reversible neutropenia (leukopenia). From the side of the central nervous system: peripheral neuropathy (feeling of numbness of the extremities), rarely and only with long-term use, headaches, convulsions, drowsiness, dizziness, loss of coordination, ataxia, confusion occur. Allergic reactions: skin rash, urticaria, skin itching, erythema multiforme, angioedema and anaphylactic reaction were very rare. Effect of the drug on the liver: increased activity of liver enzymes, cholestasis, and jaundice have been rarely reported. Others: fever, darkening of urine (causes a metabolite of metronidazole, has no clinical significance). These side effects are observed very rarely due to the low concentration of metronidazole in the blood during intravaginal use of Ginocaps Biforte vaginal capsules. Miconazole nitrate is not absorbed after insertion of Ginocaps Biforte vaginal capsules into the vagina. If side effects occur, treatment with Ginocaps Biforte should be discontinued.
Storage conditions
Store in a place protected from moisture and light at a temperature of 15 °C to 25 °C. Store out of the reach of children. Shelf life: 18 months. Do not use after the expiration date stated on the packaging.
Buy Ginocaps BIFORT caps.vag.200mg/750mg in container pack No. 5x2 in the pharmacy
Price for Ginocaps BIFORT caps.vag.200mg/750mg in container pack No. 5x2
Instructions for use for Ginocaps BIFORT caps.vag.200mg/750mg in container pack No. 5x2
Gynocaps caps vag 100 mg+100 mg x10
ATX code: G01AF20 (Combinations of imidazole derivatives)
Active substances
metronidazole (metronidazole) Rec.INN registered by WHO miconazole (miconazole) Rec.INN registered by WHO
Dosage form
Gynokaps
Vaginal capsulesreg. No.: LP-004061 dated 12/29/16 - Valid
Release form, packaging and composition of the drug Ginocaps
Vaginal capsules are soft, gelatinous, egg-shaped, with a pointed end, with a seam, elastic, opaque, light brown in color, the contents of the capsules are an oil suspension from light yellow to dark yellow with white inclusions and a non-rancid odor, delamination is possible during storage mixtures.
1 tab.
metronidazole 100 mg
miconazole nitrate 100 mg
Excipients: purified lecithin - 45 mg, beeswax - 144 mg, purified water - 108 mg, sunflower oil - a sufficient amount to obtain the contents of the capsule weighing 1800 mg.
Composition of the gelatin capsule shell: gelatin - 268 mg, glycerol - 134 mg, purified water - 35 mg, methyl parahydroxybenzoate E-218 - 0.5 mg, propyl parahydroxybenzoate E-216 - 0.13 mg, titanium dioxide E-171 - 1.8 mg, dye: "solar" sunset" yellow E-110 - 0.026 mg.
Clinical-pharmacological group: Drug with antibacterial, antiprotozoal and antifungal action for topical use in gynecology Pharmaco-therapeutic group: Antimicrobial combined agent (antimicrobial and antiprotozoal agent + antifungal agent)
pharmachologic effect
Combined product for intravaginal use.
Metronidazole is an antiprotozoal and antimicrobial drug, a derivative of nitro-5-imidazole. The mechanism of action is the biochemical reduction of the 5-nitro group of metronidazole by intracellular transport proteins of anaerobic microorganisms and protozoa. The reduced 5-nitro group of metronidazole interacts with the DNA of microbial cells, inhibiting the synthesis of their nucleic acids, which leads to the death of bacteria and protozoa.
Active against Trichomonas vaginalis, Entamoeba histolytica, Gardnerella vaginalis, Giardia lamblia, as well as obligate anaerobes Bacteroides spp. (including Bacteroides fragilis, Bacteroides ovatus, Bacteroides distasonis, Bacteroides thetaiotaomicron, Bacteroides vulgatus), Fusobacterium spp., Veillonella spp., Prevotella spp. (Prevotella bivia, Prevotella buccae, Prevotella disiens) and some gram-positive microorganisms (Eubacterium spp., Clostridium spp., Peptococcus spp., Peptostreptococcus spp.). The MIC for these strains is 0.125-6.25 μg/ml.
Aerobic microorganisms and facultative anaerobes are resistant to metronidazole, but in the presence of mixed flora (aerobes and anaerobes), metronidazole acts synergistically with antibiotics effective against aerobes.
Miconazole has an antifungal effect against dermatophytes and yeast fungi. When used intravaginally, it is active mainly against Candida albicans. Miconazole inhibits the biosynthesis of ergosterol in fungi and changes the composition of other lipid components in the membrane, which leads to the death of fungal cells. Miconazole does not change the composition of normal microflora and vaginal pH.
Pharmacokinetics
When administered intravaginally, metronidazole is absorbed into the systemic circulation. The Cmax of metronidazole in the blood is determined after 6-12 hours and is approximately 50% of the Cmax that is achieved (after 1-3 hours) after a single dose of an equivalent dose of metronidazole orally. Metronidazole penetrates into breast milk and most tissues, penetrates the BBB and the placental barrier. Plasma protein binding is less than 20%. Metabolized in the liver by hydroxylation, oxidation and glucuronidation. The activity of the main metabolite (2-hydroxymetronidazole) is 30% of the activity of the parent compound. Excreted by the kidneys - 60-80% of the dose of the systemic drug (20% of this amount unchanged). The metabolite of metronidazole, 2-hydroxymetronidazole, colors urine red-brown due to the presence of a water-soluble pigment formed as a result of the metabolism of metronidazole. The intestines excrete 6-15% of the dose of the systemic drug.
Systemic absorption of miconazole after intravaginal use is low. Rapidly destroyed in the liver. It overcomes histohematic barriers poorly. 8 hours after using the drug, 90% of miconazole is still present in the vagina. Unchanged miconazole is not detectable in either plasma or urine.
Indications of the active substances of the drug Ginocaps For the local treatment of vaginitis of mixed etiology caused simultaneously by Trichomonas spp. and Candida spp. ICD-10 codes
ICD-10 code Indication
A59 Trichomoniasis
B37.3 Candidiasis of the vulva and vagina
Dosage regimen
Use intravaginally in 1 single dose daily for 10 days. Can be used in combination with metronidazole for oral administration.
Side effect
Local reactions: itching, burning, pain, irritation of the vaginal mucosa, thick, white, mucous vaginal discharge without odor or with a faint odor, burning sensation or irritation of the partner’s penis.
From the digestive system: nausea, vomiting, changes in taste, metallic taste in the mouth, decreased appetite, spastic abdominal pain, diarrhea, constipation.
From the side of the central nervous system: headache, dizziness.
From the hematopoietic system: leukopenia or leukocytosis.
Allergic reactions: urticaria, itching of the skin, rash.
From the urinary system: frequent urination, urine coloration in red-brown color due to the presence of a water-soluble pigment (metronidazole metabolite - 2 oxymetronidazole), formed as a result of the metabolism of metronidazole.
Contraindications for use
Leukopenia (including a history), organic lesions of the central nervous system (including epilepsy), liver failure, first trimester of pregnancy, lactation period (breastfeeding), children under 18 years of age, hypersensitivity to the components of the drug and to other azoles.
Carefully
Diabetes mellitus, microcirculation disorders.
Use during pregnancy and breastfeeding
Use in the first trimester of pregnancy is contraindicated. The use of the drug in the second and third trimesters is possible only in cases where the potential benefit to the mother outweighs the risk to the fetus.
Metronidazole is excreted in breast milk. Use is contraindicated during breastfeeding. If necessary, use during lactation should stop breastfeeding.
Use for liver dysfunction Contraindicated in liver failure (when prescribing the drug in high doses).
Use in children Use in children under 18 years of age is contraindicated.
special instructions
During the use of this combination, alcohol consumption is strictly prohibited (risk of developing disulfiram-like reactions).
During the period of use of the drug, it is recommended to abstain from sexual intercourse. Simultaneous treatment of sexual partners is recommended.
During treatment, slight leukopenia is possible, so it is advisable to monitor the blood picture (the number of leukocytes) at the beginning and at the end of therapy.
Metronidazole can immobilize treponemes, resulting in a false-positive TPI test (Nelson test).
Impact on the ability to drive vehicles and machinery
If side effects from the central nervous system occur, you should refrain from driving vehicles and working with potentially dangerous mechanisms.
Drug interactions
Since the systemic absorption of miconazole is low, interactions with other drugs are caused by metronidazole.
Metronidazole is compatible with sulfonamides and antibiotics.
When consumed simultaneously, alcohol causes reactions similar to disulfiram (cramping abdominal pain, nausea, vomiting, headache, skin flushing). Concomitant use with disulfiram is unacceptable (additive effects, may cause confusion).
The drug can enhance the effect of indirect anticoagulants. Prothrombin time may increase, so dose adjustment of indirect anticoagulants is necessary.
It is not recommended to combine the drug with non-depolarizing muscle relaxants (vecuronium bromide).
Inducers of microsomal oxidation enzymes in the liver (for example, phenytoin, phenobarbital) can accelerate the elimination of metronidazole, which leads to a decrease in its concentration in the blood plasma.
Cimetidine inhibits the metabolism of metronidazole, which may lead to an increase in its concentration in the blood serum and an increased risk of side effects.
It is possible that the concentration of lithium in the blood plasma may increase during the course of treatment with metronidazole, therefore, before starting the use of this combination, it is necessary to reduce the dose of lithium or stop taking it for the duration of treatment.
Current issues of women's health. Gynokaps line
Every woman has encountered inflammatory diseases of the genitourinary system at least once in her life.
Gynecological diseases often occur against the background of untreated inflammation. Let us dwell in more detail on the treatment of infectious and inflammatory diseases of the female genital area. In the treatment of almost all inflammatory diseases of the female genital area, local treatment is used in combination with the main prescriptions: vaginal capsules, vaginal suppositories and vaginal tablets. Local forms, once in the vaginal cavity, release active substances that directly affect the foci of infection/inflammation, and, being absorbed into the tissues and entering the bloodstream, have an anti-inflammatory, antimicrobial effect.
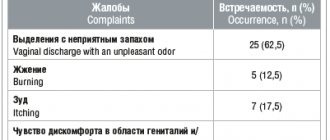
Abnormal vaginal discharge syndrome most often accompanies diseases such as bacterial vaginosis, vulvovaginal candidiasis and trichomoniasis. Bacterial vaginosis accounts for up to 87% of cases of pathological vaginal discharge in women of childbearing age. In general, in the population, bacterial vaginosis affects from 16 to 65% of women; the frequency of bacterial vaginosis in pregnant women is 15–37%.
In addition to pathological symptoms, bacterial vaginosis is dangerous because it can cause serious gynecological and obstetric complications: postpartum endometritis, complications after abortion and gynecological operations, as well as inflammatory diseases of the pelvic organs. In pregnant women, bacterial vaginosis can cause premature birth, intra-amniotic infection and, as a consequence, prenatal fetal death.
Currently, Minskintercaps UE, within the framework of the “Women’s Health” direction, produces GINOKAPS
,
GINOCAPS FORTE
and
GINOCAPS BIFORT
in the form
of vaginal capsules
.
These drugs are a fixed combination of metronidazole and miconazole for intravaginal use in various dosages, which has antibacterial, antiprotozoal and antifungal effects.
Thanks to the active substances, the Ginocaps line of drugs is effective in the treatment of common infectious and inflammatory diseases of the genitourinary tract in women - namely bacterial vaginosis, vulvovaginal candidiasis, mixed vaginal infections.
Drug components – metronidazole
and
miconazole
are as close to each other in pharmacokinetics as possible, which is important in the treatment of mixed vaginal infections; Metronidazole is active against Trichomonas and other protozoan microorganisms.
The use of a local form allows you to significantly reduce the dose of the drug or completely avoid systemic therapy. This reduces the number of side effects and allows metronidazole to be prescribed to patients with a complicating medical history.
Main indications for use of Gynocaps:
• treatment of mixed vaginal infections: bacterial vaginosis in combination with vulvovaginal candidiasis; • treatment of recurrent vaginitis and vaginitis resistant to other treatment (Ginocaps forte, Ginocaps Biforte); • treatment of trichomoniasis.
Medicines GINOKAPS BIFORTE, GINOKAPS FORTE, GINOKAPS ARE CONVENIENT TO USE: soft capsules are easily inserted into the vagina even with severe inflammation, the active substances are quickly released, evenly distributed throughout the entire mucosa and have a therapeutic effect. When treating vaginitis, swelling, discomfort and itching are quickly eliminated.
Here are the main advantages and distinctive features of the vaginal capsule dosage form:
1. Ease of storage.
Vaginal capsules retain their shape well at elevated temperatures (up to 25 ° C); there is no need to store the drug in the refrigerator.
Vaginal suppositories are unstable at room temperature and require refrigeration. 2. Ease of administration.
The vaginal capsule has a streamlined teardrop shape.
Insertion into the vagina occurs with the blunt end of the capsule, which, if necessary, is moistened with water. It is worth noting the advantage of vaginal capsules in comparison with vaginal suppositories in this parameter. Typically, the melting point of vaginal suppositories is about 35 °C. Upon contact with hands, suppositories may melt slightly, leaving marks. Vaginal capsules have a different mechanism for releasing the active contents and soften at a temperature of about 37 ° C. The time of contact with the hands does not allow the vaginal capsule to soften; it remains sufficiently hard for insertion. 3. The mechanism of release of active substances.
The vaginal capsule consists of a shell and a filler, which can be in the form of a solution, viscous suspension or emulsion.
During the production of vaginal capsules, a drying process is carried out, which leads to excess pressure inside the capsule. Under the influence of body temperature, the capsule breaks at the seam within 2-5 minutes, and the active substances are released. This mechanism contributes to the rapid onset of the therapeutic effect when using vaginal capsules. 4. Modern production technologies.
An additional advantage of vaginal capsules is the possibility of using a filler in the form of a microsuspension, microemulsion, which is not possible in the case of suppositories.
The technological capabilities for the production of vaginal capsules containing various active substances are quite wide and exceed those for the production of suppositories. 5. Security.
All components of vaginal capsules are approved for use from a safety point of view. The auxiliary components used in production are non-toxic, do not have a local irritant effect, do not cause mutagenic, carcinogenic, embryotoxic effects, etc.
The GINOCAPS line is:
GINOCAPS BIFORTE, vaginal capsules - metronidazole 750 mg / miconazole 200 mg; GINOCAPS FORTE, vaginal capsules – metronidazole 500 mg/miconazole 100 mg; GINOCAPS, vaginal capsules – metronidazole 100 mg/miconazole 100 mg.
Let's take a closer look at each drug.
GINOCAPS BIFORT
Composition per vaginal capsule: metronidazole 750 mg/miconazole 200 mg. Indications and dosage regimen: Trichomoniasis: 1 capsule 1 time/day in the evening before bed for 10 days in combination with oral metronidazole; Bacterial vaginosis: 1 capsule 1-2 times/day for 10 days, if necessary - in combination with oral metronidazole; Vulvovaginal candidiasis: 1 capsule 1 time per day for 10 days. Recurrent vaginitis or vaginitis resistant to other treatment: 1 capsule at night and 1 capsule in the morning for 14 days.
GINOCAPS FORTE
Composition per vaginal capsule: metronidazole 500 mg/miconazole 100 mg. Indications and dosage regimen: Trichomoniasis: 1 capsule 1 time/day in the evening before bed for 10 days in combination with oral metronidazole; Bacterial vaginosis: 1 capsule 1-2 times/day for 10 days, if necessary - in combination with oral metronidazole; Vulvovaginal candidiasis: 1 capsule 1 time per day for 10 days. Recurrent vaginitis or vaginitis resistant to other treatment: 1 capsule at night and 1 capsule in the morning for 14 days.
GYNOCAPS
Composition per vaginal capsule: metronidazole 100 mg/miconazole 100 mg. Indications and dosage regimen: bacterial vaginosis: 1 capsule 1-2 times a day for 10 days; trichomoniasis: 1 capsule 1 time per day for 10 days; candidal vaginitis (thrush): 1 capsule once a day for 10 days; mixed vaginal infections: 1 capsule 1 time per day for 10 days.
Ginocaps is the optimal choice for patients in the treatment of mixed vaginal infections and an ambulance for women's health! It should be remembered that inflammatory processes in the vagina are a serious intimate problem that can provoke the development of irreversible consequences, so you should not put off visiting a doctor. Ginocaps is an optimal alternative to the systemic treatment of vaginal infections. Ginocaps is confidence in effectiveness and safety!
BEFORE USING MEDICINES, READ THE INSTRUCTIONS AND CONSULT A DOCTOR. There are medical contraindications and adverse reactions. Use is contraindicated in the first trimester of pregnancy.
Find out where to buy and how much the drug Ginocaps
,
Ginocaps Forte
and
Ginocaps Biforte
can be found
here
.
26 Jul 2021
Previous News Feed Next