Description of drug types
Desal is available in the form of tablets and oral solution. The main active ingredient in both cases is desloratadine.
The tablets have a blue film shell and a round, biconvex shape. One piece contains 5 mg of desloratadine. Additionally, the composition includes: magnesium stearate, cellulose, corn starch and mannitol.
The tablets are packaged in cell plates of 10 pieces, and go on sale in cardboard boxes of 10 or 30 pieces.
The liquid form is presented as a clear, colorless solution with a fruity aroma and sour taste; 1 ml contains 0.5 mg of desloratadine. Sold in 100 ml bottles complete with a measuring syringe and instructions, which are placed in a box.
Desal solution for oral administration 0.5 mg/ml bottle 100 ml
A country
Bulgaria
The country of production may vary depending on the batch of goods. Please check with the operator for detailed information when confirming your order.
Active substance
Desloratadine
Compound
A bottle of 100 billion -zloratadine 0.5 mg per 1 ml. Auxiliary substances: sorbitol - 147.15 mg, propylene glycol - 102.3 mg, citric acid monohydrate - 21.06 mg, sodium citrate dihydrate - 16.38 mg, gypromellose 2910 - 2 mg, 1 mg, dinatri Edetta - dinatriy 0.04 mg, tutti-frutti flavoring - 0.03 mg. Oral solution is clear, colorless, free from foreign particles.
pharmachologic effect
Long-acting antihistamine, peripheral histamine H1 receptor blocker. Desloratadine is the primary active metabolite of loratadine. Inhibits the cascade of allergic inflammation reactions, incl. release of proinflammatory cytokines, including interleukins IL-4, IL-6, IL-8, IL-13, release of proinflammatory chemokines, production of superoxide anions by activated polymorphonuclear neutrophils, adhesion and chemotaxis of eosinophils, release of adhesion molecules such as P-selectin, IgE- mediated release of histamine, prostaglandin D2 and leukotriene C4. Thus, the drug prevents the development and facilitates the course of allergic reactions, has an antipruritic and anti-exudative effect, reduces capillary permeability, prevents the development of tissue edema, spasm of smooth muscles. The drug has no effect on the central nervous system, has virtually no sedative effect (does not cause drowsiness) and does not affects the speed of psychomotor reactions when taken in recommended doses. Does not cause prolongation of the QT interval on the ECG. The action of desloratadine begins within 30 minutes after oral administration and continues for 24 hours.
Indications for use
To relieve or eliminate the symptoms of: - allergic rhinitis (sneezing, nasal congestion, rhinorrhea, itchy nose, itchy palate, itching and redness of the eyes, lacrimation); - urticaria (itchy skin, rash).
Mode of application
Take orally, regardless of food intake. Children aged 1 to 5 years - 2.5 ml of solution (1.25 mg) 1 time / day. Children aged 6 to 11 years - 5 ml of solution (2.5 mg) 1 time / day. Adults and adolescents (12 years and older) - 10 ml of solution (5 mg) 1 time / day.
Interaction
No clinically significant interactions with other drugs have been identified (including ketoconazole and erythromycin). Desloratadine does not enhance the effect of ethanol on the central nervous system.
Side effect
In children aged 2 to 11 years, when using desloratadine, the frequency of side effects was the same as when using placebo. In children under 2 years of age, when using desloratadine, the following side effects were observed, the frequency of which was slightly higher than when using placebo: diarrhea (3.7%), increased body temperature (2.3%), insomnia (2.3%). In adults and adolescents aged 12 years and older, the following side effects were observed when using desloratadine, the frequency of which was slightly higher than when using placebo: increased fatigue (1.2%), dry mouth (0.8%), headache (0.6%). When using the drug in adults and adolescents at the recommended dose of 5 mg / day, the incidence of drowsiness is not higher than when using placebo. During post-marketing surveillance the following adverse reactions have been very rarely observed. Mental disorders: hallucinations. From the central nervous system: dizziness, drowsiness, insomnia, psychomotor hyperactivity. From the cardiovascular system: tachycardia, palpitations. From the digestive system: abdominal pain, nausea, vomiting, dyspepsia, diarrhea. From the liver and biliary tract: increased activity of liver enzymes, increased concentration of bilirubin, hepatitis. From the musculoskeletal system: myalgia. Allergic reactions: anaphylaxis, angioedema, itching, rash, urticaria.
Contraindications
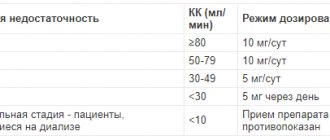
- hypersensitivity to the active or any excipient of the drug; - pregnancy; - lactation period (breastfeeding); - children under 1 year of age (efficacy and safety have not been established); - hereditary diseases - fructose intolerance, glucose/galactose malabsorption or deficiency of sucrase/isomaltase in the body (due to the presence of sorbitol in the drug). The drug should be used with caution in severe renal failure.
Overdose
Symptoms: taking a dose 9 times higher than the recommended dose (45 mg) did not lead to the appearance of any clinically significant symptoms. Drowsiness may develop. Treatment: gastric lavage is necessary, taking activated charcoal; if necessary, symptomatic therapy. Desloratadine is not eliminated by hemodialysis; the effectiveness of peritoneal dialysis has not been established. If a large amount of the drug is accidentally ingested, the patient should immediately consult a doctor.
special instructions
The effectiveness and safety of the drug Dezal in children under 1 year of age has not been established. Differential diagnosis between allergic rhinitis and rhinitis of other origin in children under 2 years of age presents certain difficulties. The differential diagnosis should include consideration of the presence or absence of foci of infection or structural abnormalities of the upper respiratory tract, a thorough history, examination, and appropriate laboratory studies and skin tests. In approximately 6% of adults and children aged 2 to 11 years, there is a low ability of desloratadine to metabolize; desloratadine is excreted more slowly in this group of patients. The safety profile of desloratadine in children aged 2 to 11 years with low metabolic rates is similar to that in children with normal metabolism of desloratadine. The effect of desloratadine on children under 2 years of age with a low metabolic rate has not been studied. In cases of severe renal impairment, Desal should be taken with caution. Effects on the ability to drive vehicles and machinery In studies of the effect of desloratadine on the ability to drive vehicles, no impairment of concentration was observed. However, it should be borne in mind that very rarely some patients may develop drowsiness; in this case, care should be taken when driving vehicles and when working with machinery.
How does Dezal work?
The medication belongs to the group of antihistamine receptor blockers, that is, it is intended to suppress a chain of allergic reactions. It acts in the following areas:
- prevents the development of allergy symptoms;
- improves the condition of allergies;
- relieves itching;
- prevents the development of tissue swelling;
- reduces mucus secretion and lacrimation;
- strengthens the walls of small blood vessels;
- serves as a prevention of spasms.
Desal tablets do not inhibit the functioning of the nervous system, have a minimal sedative effect, and if the dosage regimen is followed, they do not provoke lethargy and do not impair the speed of reaction.
The effect of the tablet begins half an hour after taking it. The effect lasts throughout the day.
Desal 5mg No. 10 tablets
Release form
Pills. Pack of 10 pcs.
pharmachologic effect
Desal is an antiallergic drug - a blocker of histamine H1 receptors. Long-acting antihistamine, peripheral histamine H1 receptor blocker. Desloratadine is the primary active metabolite of loratadine. Inhibits the cascade of allergic inflammation reactions, incl. release of proinflammatory cytokines, including interleukins IL-4, IL-6, IL-8, IL-13, release of proinflammatory chemokines, production of superoxide anions by activated polymorphonuclear neutrophils, adhesion and chemotaxis of eosinophils, release of adhesion molecules such as P-selectin, IgE- mediated release of histamine, prostaglandin D2 and leukotriene C4. Thus, it prevents the development and facilitates the course of allergic reactions, has antipruritic and antiexudative effects, reduces capillary permeability, and prevents the development of tissue edema and smooth muscle spasm. The drug has no effect on the central nervous system, has virtually no sedative effect (does not cause drowsiness) and does not affect the speed of psychomotor reactions when taken in recommended doses. Does not cause prolongation of the QT interval on the ECG. The action of desloratadine begins within 30 minutes after oral administration and continues for 24 hours.
Indications
To relieve or eliminate symptoms of: allergic rhinitis (sneezing, nasal congestion, rhinorrhea, itchy nose, itchy palate, itchy and red eyes, watery eyes); urticaria (skin itching, rash).
Contraindications
hypersensitivity to the active or any excipient of the drug; - pregnancy; - period of breastfeeding; - age up to 12 years (efficacy and safety have not been established); The drug should be used with caution in severe renal failure.
Use during pregnancy and breastfeeding
Prescribing the drug during pregnancy is not recommended due to the lack of clinical data on the safety of its use during this period. Desloratadine is excreted in breast milk, so its use during breastfeeding is not recommended.
special instructions
In case of severe renal impairment, Dezal should be taken with caution. Studies have shown no effect of desloratadine on driving. However, it should be borne in mind that very rarely some patients may develop drowsiness; in this case, care should be taken when driving vehicles and when working with machinery.
Compound
1 tablet contains: Active substance: desloratadine 5 mg; Excipients: microcrystalline cellulose, pregelatinized corn starch, mannitol, talc, magnesium stearate
Directions for use and doses
Inside, regardless of food intake. Adults and adolescents (12 years and older) - 5 mg (1 tablet) 1 time per day.
Side effects
The following adverse reactions are most often observed: increased fatigue (1.2%), dry oral mucosa (0.8%), headache (0.6%). When using the drug in adults and adolescents at the recommended dose of 5 mg/day, the incidence of drowsiness is not higher than when using placebo. During post-marketing surveillance, the following adverse reactions were very rarely observed. Mental disorders: hallucinations. From the central nervous system: dizziness, drowsiness, insomnia, psychomotor hyperactivity. From the cardiovascular system: tachycardia, palpitations. From the digestive system: abdominal pain, nausea, vomiting, dyspepsia, diarrhea. From the liver and biliary tract: increased activity of liver enzymes, increased bilirubin concentration, hepatitis. From the musculoskeletal system: myalgia. Allergic reactions: anaphylaxis, angioedema, itching, rash, urticaria.
Drug interactions
No clinically significant interactions with other drugs have been identified (including ketoconazole and erythromycin). Desloratadine does not enhance the effect of ethanol on the central nervous system.
Overdose
Symptoms: taking a dose 9 times higher than the recommended dose (45 mg) did not lead to the appearance of any clinically significant symptoms. Drowsiness may develop. Treatment: gastric lavage is necessary, taking activated charcoal; if necessary, symptomatic therapy. Desloratadine is not eliminated by hemodialysis; the effectiveness of peritoneal dialysis has not been established.
Storage conditions
Store in a dry place, protected from light, out of reach of children at a temperature not exceeding 25°C.
Best before date
2 years.
Contraindications, unwanted effects
Tablets are not prescribed:
- pregnant and lactating women;
- children under 12 years old;
- persons with hypersensitivity to any of the constituent substances.
The solution is additionally contraindicated:
- infants of the first year of life;
- for congenital metabolic disorders - glucose-galactose deficiency, fructose intolerance, since sorbitol is present in the composition.
Severe kidney damage is a reason for careful use of the medicine.
As a rule, the drug is well tolerated. In some cases, it can cause excessive fatigue, a feeling of dryness of the mucous membranes of the oral cavity, and pain in the head.
Very rare negative effects such as hallucinations, sleep disturbances/drowsiness, increased heart rate, nervous system excitement, and dizziness are reported.
May cause nausea, vomiting, diarrhea and abdominal discomfort and muscle weakness. In case of intolerance - Quincke's edema, anaphylactic shock, rashes and itching.
The manufacturer warns that during studies, a small number of children under two years of age experienced adverse reactions such as diarrhea, hyperthermia and sleep disturbances.
Dezal
In children aged 6 to 23 months, the following side effects were observed, the frequency of which was slightly higher than when using placebo (“dummies”): diarrhea (in 3.7% of cases), increased body temperature (2.3%), insomnia (2.3%).
In children aged 2 to 11 years, the incidence of side effects with desloratadine was the same as with placebo.
According to the results of clinical studies in children aged 6 to 11 years, no side effects were identified when taking the drug in recommended doses (2.5 mg/day).
In children aged 12-17 years, according to the results of clinical studies, the most common side effect was headache (5.9%), the frequency of which was higher than with placebo (6.9%).
In adults and adolescents (12 years of age and older), according to the results of clinical studies, side effects were recorded in 3% of patients compared to the placebo group, the most common of which were: increased fatigue (1.2%), dry mouth (0). .8%), headache (0.6%).
Information on side effects is presented based on the results of clinical studies and post-registration observations.
According to the World Health Organization (WHO), side effects are classified according to their frequency as follows: very common (≥1/10), common (≥1/100 to <1/10), uncommon (≥1/1000 to <1/100), rare (from ≥1/10,000 to <1/1000), very rare (<1/10,000), frequency unknown - it was not possible to determine the frequency of occurrence based on available data.
Metabolism and nutrition:
frequency unknown - increased appetite. From the mental side: very rarely - hallucinations; frequency unknown abnormal behavior, aggression.
From the nervous system
: often - headache; often (in children under 2 years of age) - insomnia; very rarely - dizziness, drowsiness, insomnia, psychomotor hyperactivity, convulsions.
From the liver and biliary tract:
very rarely - increased activity of liver enzymes, increased bilirubin concentration, hepatitis; frequency unknown - jaundice.
From the digestive system:
often - dry mouth, often (in children under 2 years of age) - diarrhea; very rarely - abdominal pain, nausea, vomiting, dyspepsia, diarrhea.
From the cardiovascular system:
very rarely - tachycardia, palpitations; frequency unknown; QT interval prolongation.
From the musculoskeletal system:
very rarely - myalgia.
For the skin and subcutaneous tissues:
frequency unknown - photosensitivity.
Common disorders:
often - increased fatigue, often (in children under 2 years of age) - fever; very rarely - anaphylaxis, angioedema, shortness of breath, itching, rash, including urticaria; frequency unknown - asthenia.
Laboratory and instrumental data:
frequency unknown - weight gain.
Post-registration period
Children
: frequency unknown - QT prolongation, arrhythmia, bradycardia, abnormal behavior, aggression.
If any of the side effects indicated in the instructions get worse, or you notice any other side effects not listed in the instructions, tell your doctor.
Important instructions
If it is necessary to operate transport or complex mechanisms, it is necessary to take into account the individual characteristics of the body. In some cases, the drug can cause drowsiness and lethargy, so you need to be careful during hazardous activities.
In case of overdose, severe drowsiness is possible. In this case, it is recommended to rinse the stomach and take activated charcoal. If necessary, symptomatic treatment is prescribed.
Significant interactions with other pharmaceuticals, such as the antibiotic azithromycin, have not been established.
Reviews, analogues
Doctors' opinions on the Internet about Dezale are mostly positive. The convenience of a single dose, a variety of dosage forms, good tolerability, and speed of action are noted. The drug also helps to reduce swelling in viral diseases of the ENT organs.
Buyers point out the acceptable cost, long-lasting effect, modernity of the drug and lack of addiction as advantages.
Analogues of the active substance include: Erius, Lordestin, Blogir-3, Desloratadine tablets, Ezlor Solution Tablets. The following new generation drugs also have an antihistamine effect: Suprastinex, Zodak Express based on levocetirizine, Allegra and Telfast based on fexofenadine.