Relevance
Invasive aspergillosis is the most common invasive infection after organ transplantation and occurs in 1-15% of recipients. The 12-week mortality rate ranges from 20 to 60%. The highest risk of the disease is observed after lung transplantation. In addition, Aspergillus colonization is an independent risk factor for chronic lung graft dysfunction. All things considered, most lung transplant patients receive prophylactic antifungal therapy.
Special instructions for the use of the drug Voriconazole
Use with caution in patients with severe liver failure, with severe renal failure (with parenteral administration), as well as with hypersensitivity to other drugs - azole derivatives. Before starting treatment, correction of electrolyte disturbances (hypokalemia, hypomagnesemia and hypocalcemia) is required. Sampling for cultural and other laboratory tests (serological, histopathological) for the purpose of isolating and identifying pathogens should be done before treatment. Therapy can be started pending laboratory results and then adjusted if necessary. The use of voriconazole may lead to prolongation of the QT on the ECG, which is accompanied by rare cases of ventricular fibrillation/flutter in patients with multiple risk factors (cardiotoxic chemotherapy, cardiomyopathy, hypokalemia and concomitant medications that could contribute to the development of adverse events from the cardiovascular system ). Voriconazole should be used with caution in patients with these potentially proarrhythmic conditions. During the treatment period, liver function should be regularly monitored (if clinical signs of liver disease appear, the advisability of discontinuing therapy should be discussed), kidney function (including serum creatinine level). If dermatological reactions progress, the drug should be discontinued. During treatment, patients receiving voriconazole should avoid exposure to the sun and UV radiation. With the simultaneous use of voriconazole in patients receiving cyclosporine and tacrolimus, the dose of the latter should be adjusted and their concentration in the blood plasma should be monitored. After discontinuation of voriconazole, the plasma concentrations of cyclosporine and tacrolimus should be assessed and their dose increased if necessary. If concomitant use of voriconazole and phenytoin is necessary, the expected benefits and potential risks of combination therapy should be carefully assessed and phenytoin levels should be continuously monitored. If it is necessary to use voriconazole and rifabutin in combination, the expected benefits and potential risks of combination therapy should be carefully assessed and carried out under the supervision of peripheral blood patterns, as well as other possible undesirable effects of rifabutin. The safety and effectiveness of voriconazole in children under 2 years of age have not been established. Influence on the ability to drive vehicles and operate machinery. Because voriconazole may cause transient visual disturbances, including blurred vision, blurred/increased visual perception and/or photophobia, if such reactions occur, patients should not engage in potentially hazardous activities, such as driving or using complex machinery. While using voriconazole, patients should not drive in the dark. Use during pregnancy and lactation. Adequate and strictly controlled studies on the safety of voriconazole during pregnancy have not been conducted. Experimental studies on animals have shown that voriconazole in high doses has a toxic effect on reproductive function. The possible risk to humans is unknown. The excretion of voriconazole into breast milk has not been studied. Voriconazole should not be used during pregnancy or breastfeeding unless the expected benefit to the mother outweighs the potential risk to the fetus or infant. During treatment, women of reproductive age should use reliable methods of contraception.
Safety of voriconazole
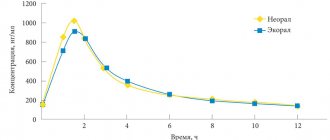
Currently, the drug of choice is voriconazole. Although the results of a meta-analysis indicate that the use of voriconazole significantly reduces the risk of disabling aspergillosis, the safety of therapy raises questions. In particular. Voriconazole has multiple drug interactions due to the induction of cytochrome P450: it increases serum levels of calcium neuron inhibitors and affects the concentration of rapamycin (mTOR).
- Known side effects of the antifungal drug also include blurred vision, liver and kidney failure, and increased skin photosensitivity.
- In addition, voriconazole is associated with an increased risk of squamous cell skin cancer, which is reflected in the instructions for the drug.
Description of the drug VORICONAZOLE (VORICONAZOLE)
Voriconazole is metabolized with the participation of isoenzymes CYP2C19, CYP2C9 and CYP3A4. Inhibitors or inducers of these isoenzymes may cause an increase or decrease in voriconazole plasma concentrations, respectively.
When used simultaneously with rifampicin (an inducer of CYP isoenzymes) at a dose of 600 mg/day, the Cmax and AUC of voriconazole are reduced by 93% and 96%, respectively (the combination is contraindicated).
When coadministered with voriconazole, ritonavir (a CYP isoenzyme inducer, CYP3A4 inhibitor and substrate) at a dose of 400 mg every 12 hours reduced the steady-state Cmax and AUC of oral voriconazole by an average of 66% and 82%, respectively. The effect of lower doses of ritonavir on voriconazole concentrations is not yet known. It has been established that repeated oral use of voriconazole does not have a significant effect on Cmax at steady state and AUC of ritonavir, also taken repeatedly (simultaneous use of voriconazole and ritonavir at a dose of 400 mg every 12 hours is contraindicated).
When used concomitantly with potent CYP isoenzyme inducers carbamazepine or long-acting barbiturates (phenobarbital), a significant decrease in voriconazole plasma Cmax is possible, although their interaction has not been studied. This combination is contraindicated.
When used together with cimetidine (a nonspecific inhibitor of CYP isoenzymes) at a dose of 400 mg 2 times / day, the Cmax and AUC of voriconazole increase by 18% and 23%, respectively (no dose adjustment of voriconazole is required).
Voriconazole inhibits the activity of CYP2C19, CYP2C9, CYP3A4, so it is possible to increase plasma concentrations of drugs that are metabolized by these isoenzymes.
With the simultaneous use of voriconazole with terfenadine, astemizole, cisapride, pimozide and quinidine, a significant increase in their plasma concentrations is possible, which can lead to prolongation of the QT interval and in rare cases to the development of ventricular fibrillation/flutter (the combination is contraindicated).
When used together, voriconazole increases the Cmax and AUC of sirolimus (2 mg once) by 556% and 1014%, respectively (the combination is contraindicated).
When used concomitantly, voriconazole may cause an increase in plasma concentrations of ergot alkaloids (ergotamine and dihydroergotamine) and the development of ergotism (this combination is contraindicated).
When used together in stable renal transplant patients, voriconazole increases the Cmax and AUC of cyclosporine by at least 13% and 70%, respectively, which is accompanied by an increased risk of nephrotoxic reactions. When using voriconazole in patients receiving cyclosporine, it is recommended to reduce the dose of cyclosporine by 2 times and monitor its plasma levels. After discontinuation of voriconazole, cyclosporine levels should be monitored and the dose increased if necessary.
When used together, voriconazole increases the Cmax and AUC of tacrolimus (used at a dose of 0.1 mg/kg once) by 117% and 221%, respectively, which may be accompanied by nephrotoxic reactions. When using voriconazole in patients receiving tacrolimus, it is recommended to reduce the dose of the latter to 1/3 and monitor its plasma levels. After discontinuation of voriconazole, it is necessary to monitor the concentration of tacrolimus and, if necessary, increase its dose.
The simultaneous use of voriconazole (at a dose of 300 mg 2 times / day) and warfarin (30 mg 1 time / day) was accompanied by an increase in the maximum prothrombin time to 93%. When prescribing warfarin and voriconazole simultaneously, it is recommended to monitor prothrombin time.
Voriconazole, when used together, may cause an increase in plasma concentrations of phenprocoumon, acenocoumarol (CYP2C9, CYP3A4 substrates) and prothrombin time. When using voriconazole in patients receiving coumarin drugs, it is necessary to monitor prothrombin time at short intervals and adjust the dose of anticoagulants accordingly.
When used together, voriconazole may increase plasma concentrations of sulfonylurea derivatives (CYP2C9 substrates) tolbutamide, glipizide and glibenclamide and cause hypoglycemia. If they are used concomitantly, carefully monitor blood glucose levels.
In vitro, voriconazole inhibits the metabolism of lovastatin (a CYP3A4 substrate). When used together, it is possible to increase the plasma concentration of statins metabolized by CYP3A4, which may increase the risk of developing rhabdomyolysis. When using them simultaneously, it is recommended to evaluate the feasibility of adjusting the statin dose.
In vitro, voriconazole inhibits the metabolism of midazolam (a CYP3A4 substrate). When used together, it is possible to increase the plasma concentration of benzodiazepines metabolized under the influence of CYP3A4 (midazolam, triazolam, alprazolam) and the development of a prolonged sedative effect. If these drugs are used concomitantly, it is recommended to discuss the advisability of adjusting the benzodiazepine dose.
When used together, voriconazole may increase the content of vinca alkaloids (CYP3A4 substrates) - vincristine, vinblastine in plasma and lead to the development of neurotoxic reactions. It is recommended to discuss the appropriateness of dosage adjustments of vinca alkaloids.
Voriconazole increases the Cmax and AUC of prednisolone (CYP3A4 substrate) administered at a dose of 60 mg once by 11% and 34%, respectively. Dose adjustment is not recommended.
When used concomitantly with voriconazole, efavirenz (a CYP3A4 substrate, according to a number of studies, depending on the dose - an inhibitor or inducer of CYP3A4), used at a dose of 400 mg 1 time / day at steady state, reduces the Cmax and AUC of voriconazole by an average of 61% and 77 % respectively. Voriconazole at steady state (400 mg PO every 12 hours on day 1, then 200 mg PO every 12 hours for 8 days) increased the steady-state Cmax and AUC of efavirenz by an average of 38% and 44%, respectively (this combination is contraindicated).
When used together, phenytoin (a CYP2C9 substrate and a powerful inducer of cytochrome P450 isoenzymes), used at a dose of 300 mg 1 time / day, reduces the Cmax and AUC of voriconazole by 49% and 69%, respectively; and voriconazole (400 mg 2 times / day) increases the Cmax and AUC of phenytoin by 67% and 81%, respectively (if coadministration is necessary, the ratio of the expected benefits and potential risks of combination therapy should be carefully assessed, and phenytoin plasma levels should be carefully monitored).
When used together, rifabutin (a cytochrome P450 inducer) used at a dose of 300 mg 1 time / day reduces the Cmax and AUC of voriconazole (200 mg 1 time / day) by 69% and 78%, respectively. When used together with rifabutin, the Cmax and AUC of voriconazole (350 mg 2 times / day) are, respectively, 96% and 68% of those with voriconazole monotherapy (200 mg 2 times / day). When using voriconazole at a dose of 400 mg 2 times / day, Cmax and AUC are, respectively, 104% and 87% higher than when using voriconazole monotherapy at a dose of 200 mg 2 times / day. Voriconazole at a dose of 400 mg 2 times / day increases the Cmax and AUC of rifabutin by 195% and 331%, respectively. During simultaneous treatment with rifabutin and voriconazole, it is recommended to regularly conduct a detailed analysis of the peripheral blood picture and monitor for undesirable effects of rifabutin (for example, uveitis).
When used together at a dose of 40 mg 1 time / day, omeprazole (inhibitor of CYP2C19; substrate of CYP2C19 and CYP3A4) increases the Cmax and AUC of voriconazole by 15% and 41%, respectively, and voriconazole increases the Cmax and AUC of omeprazole by 116% and 280%, respectively (hence , no dose adjustment of voriconazole is required, and the dose of omeprazole should be reduced by 2 times). The possibility of drug interaction between voriconazole and other H+-K+-ATPase inhibitors that are substrates of CYP2C19 should be taken into account.
When used concomitantly with other HIV protease inhibitors (substrates and inhibitors of CYP3A4), the patient's condition should be carefully monitored for possible toxic effects, because In vitro studies have shown that voriconazole and HIV protease inhibitors (saquinavir, amprenavir, nelfinavir) may mutually inhibit each other's metabolism.
Skin cancer risk
A multicenter retrospective cohort study of 900 lung transplant patients was recently performed. Approximately half of them received voriconazole for more than 30 days, and 32% of patients received another azole drug.
- According to the study results, during 3.5 years of follow-up, squamous cell carcinoma developed in 6% of patients. Multivariate analysis showed that the use of voriconazle was significantly associated with the development of skin cancer, the effect was dose-dependent. It is important to note that the association was more significant when voriconazole was prescribed for a long time (more than 180 days) and when the drug was used to prevent infection rather than treat it.
Voriconazole Canon tablets capt/vol 50mg bl N30x1 Canonpharma
Laboratory indicators: often - impaired liver function (including increased activity of AST, ALT, alkaline phosphatase, gamma-glutamyltransferase, lactate dehydrogenase, bilirubin concentration), increased concentration of creatinine in the blood plasma; uncommon – prolongation of the QT interval, increased residual urea nitrogen, hypercholesterolemia. From the cardiovascular system: very often - peripheral edema; often – decreased blood pressure, thrombophlebitis, phlebitis; uncommon – ventricular fibrillation, ventricular arrhythmia, syncope, atrial arrhythmia, supraventricular arrhythmia, supraventricular tachycardia, bradycardia, tachycardia; rarely - ventricular tachycardia (including ventricular flutter), ventricular tachysystolic arrhythmia, complete atrioventricular block, bundle branch block, nodal arrhythmias, lymphangitis. From the hematopoietic system and lymphatic system: often - pancytopenia, suppression of bone marrow hematopoiesis, thrombocytopenia, leukopenia, purpura, anemia (including: macrocytic, microcytic, normocytic, megaloblastic, aplastic); uncommon – disseminated intravascular coagulation syndrome, lymphadenopathy, agranulocytosis, eosinophilia. From the nervous system: very often – headache; often - dizziness, confusion, agitation, tremor, paresthesia; uncommon – cerebral edema, ataxia, diplopia, vertigo, hypoesthesia; rarely - convulsions, encephalopathy, Guillain-Barré syndrome, extrapyramidal disorders, drowsiness during infusion, peripheral neuropathy. On the part of the organ of vision: very often - visual disturbances (including disturbance/intensification of visual perception, the appearance of a “veil” before the eyes, changes in color perception, photophobia); uncommon – papilledema, scleritis, blepharitis, optic neuritis, nystagmus; rarely - hemorrhage into the retina of the eye, optic nerve atrophy, corneal opacification, oculogyric crisis. From the organ of hearing and vestibular apparatus: infrequently – hypoacusis, tinnitus. From the respiratory system, chest and mediastinum: often - acute respiratory distress syndrome, pulmonary edema, respiratory failure, chest pain. From the gastrointestinal tract: very often - nausea, vomiting, diarrhea, abdominal pain; uncommon – constipation, duodenitis, dyspepsia, gingivitis, glossitis, pancreatitis, swelling of the tongue, peritonitis; rarely – disturbance of taste perception. From the genitourinary system: often – acute renal failure, hematuria; uncommon – albuminuria, nephritis; rarely – necrosis of the renal tubules. From the skin and subcutaneous tissues: very often - rash; often – facial swelling, itching, maculopapular rash, macular rash, papular rash, photosensitivity, alopecia, exfoliative dermatitis, cheilitis, erythema; uncommon – Stevens-Johnson syndrome, angioedema, fixed drug rash, eczema, psoriasis, urticaria; rarely – discoid lupus erythematosus, erythema multiforme, toxic epidermal necrolysis, pseudoporphyria; frequency unknown – squamous cell carcinoma. From the musculoskeletal system and connective tissue: often – back pain; uncommon – arthritis; rarely – hypertension; frequency unknown – periostitis. From the endocrine system: infrequently – adrenal cortex insufficiency; rarely – hyperthyroidism, hypothyroidism. Metabolic and nutritional disorders: often – hypokalemia, hypoglycemia. Infections and infestations: often - gastroenteritis, influenza-like syndrome; rarely – pseudomembranous colitis. General and local reactions: very often – fever; often – chills, asthenia. From the immune system: often – sinusitis; uncommon – allergic reactions, anaphylactoid reactions. From the hepatobiliary system: often – jaundice, cholestatic jaundice; uncommon – cholecystitis, cholelithiasis, liver enlargement, hepatitis, liver failure; rarely - hepatic coma. Mental disorders: often – hallucinations, depression, anxiety; rarely – insomnia. Use in children: undesirable effects of the drug in children are similar to those in adults.